Pannexin-1 hemichannel-mediated ATP release together with P2X1 and P2X4 receptors regulate T-cell activation at the immune synapse
- PMID: 20660288
- PMCID: PMC2981474
- DOI: 10.1182/blood-2010-04-277707
Pannexin-1 hemichannel-mediated ATP release together with P2X1 and P2X4 receptors regulate T-cell activation at the immune synapse
Abstract
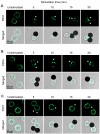
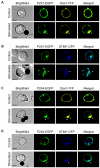
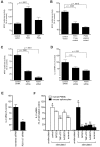
Engagement of T cells with antigen-presenting cells requires T-cell receptor (TCR) stimulation at the immune synapse. We previously reported that TCR stimulation induces the release of cellular adenosine-5'-triphosphate (ATP) that regulates T-cell activation. Here we tested the roles of pannexin-1 hemichannels, which have been implicated in ATP release, and of various P2X receptors, which serve as ATP-gated Ca(2+) channels, in events that control T-cell activation. TCR stimulation results in the translocation of P2X1 and P2X4 receptors and pannexin-1 hemichannels to the immune synapse, while P2X7 receptors remain uniformly distributed on the cell surface. Removal of extracellular ATP or inhibition, mutation, or silencing of P2X1 and P2X4 receptors inhibits Ca(2+) entry, nuclear factors of activated T cells (NFAT) activation, and induction of interleukin-2 synthesis. Inhibition of pannexin-1 hemichannels suppresses TCR-induced ATP release, Ca(2+) entry, and T-cell activation. We conclude that pannexin-1 hemichannels and P2X1 and P2X4 receptors facilitate ATP release and autocrine feedback mechanisms that control Ca(2+) entry and T-cell activation at the immune synapse.
Figures


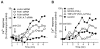
References
-
- Feske S, Gwack Y, Prakriya M, et al. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature. 2006;441(7090):179–185. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

