Balance of pro- versus anti-angiogenic splice isoforms of vascular endothelial growth factor as a regulator of neuroblastoma growth
- PMID: 20662003
- PMCID: PMC3287290
- DOI: 10.1002/path.2746
Balance of pro- versus anti-angiogenic splice isoforms of vascular endothelial growth factor as a regulator of neuroblastoma growth
Abstract
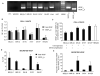
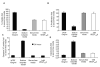
Neuroblastoma (NB) is the second most common extracranial tumour of childhood. Angiogenesis plays a crucial role in the growth and development of NB and vascular endothelial growth factor (VEGF), one of the most potent stimuli of angiogenesis, has been studied extensively in vitro. VEGF(165) has been shown to be the predominant angiogenic isoform expressed in NB cell lines and tumours. In this study, we investigated the anti-angiogenic isoform of VEGF-A, generated from distal splice site selection in the terminal exon of VEGF (VEGF(165)b) and shown to be down-regulated in epithelial malignancies. The expression of both the pro- (VEGF(xxx)) and the anti-angiogenic (VEGF(xxx)b) isoforms was compared in a range of NB and ganglioneuroma (GN) tumours. Whereas VEGF(xxx)b and VEGF(xxx) were both expressed in GN, specific up-regulation of the VEGF(xxx) isoforms was seen in NB at RNA and protein levels. Highly tumourigenic NB cell lines also showed up-regulation of the angiogenic isoforms relative to VEGF(xxx)b compared to less tumourigenic cell lines, and the isoforms were differentially secreted. These results indicate that VEGF(165) is up-regulated in NB and that there is a difference in the balance of isoform expression from anti-angiogenic VEGF(165)b to angiogenic VEGF(165). Treatment with recombinant human VEGF(165)b significantly reduced the growth rate of established xenografts of SK-N-BE(2)-C cells (4.24 +/- 1.01 fold increase in volume) compared with those treated with saline (9.76 +/- 3.58, p < 0.01). Microvascular density (MVD) was significantly decreased in rhVEGF(165)b-treated tumours (19.4 +/- 1.9 vessels/mm(3)) in contrast to the saline-treated tumours (45.5 +/- 8.6 vessels/mm(3)). VEGF(165)b had no significant effect on the proliferative or apoptotic activity, viability or cytotoxicity of SK-N-BE(2)-C cells after 48 h. In conclusion, VEGF(165)b is an effective inhibitor of NB growth. These findings provide the rationale for further investigation of VEGF(165)b in NB and other paediatric malignancies.
Copyright 2010 Pathological Society of Great Britain and Ireland. Published by John Wiley & Sons, Ltd.
Figures



Similar articles
-
The transcription factor E2F1 and the SR protein SC35 control the ratio of pro-angiogenic versus antiangiogenic isoforms of vascular endothelial growth factor-A to inhibit neovascularization in vivo.Oncogene. 2010 Sep 30;29(39):5392-403. doi: 10.1038/onc.2010.281. Epub 2010 Jul 19. Oncogene. 2010. PMID: 20639906
-
PlGF and VEGF-A Regulate Growth of High-Risk MYCN-Single Copy Neuroblastoma Xenografts via Different Mechanisms.Int J Mol Sci. 2016 Sep 23;17(10):1613. doi: 10.3390/ijms17101613. Int J Mol Sci. 2016. PMID: 27669225 Free PMC article.
-
VEGF(121)b, a new member of the VEGF(xxx)b family of VEGF-A splice isoforms, inhibits neovascularisation and tumour growth in vivo.Br J Cancer. 2009 Oct 6;101(7):1183-93. doi: 10.1038/sj.bjc.6605249. Epub 2009 Aug 25. Br J Cancer. 2009. PMID: 19707198 Free PMC article.
-
[Alternative splicing of vascular endothelial growth factor A and ocular neovascularization].Zhonghua Yan Ke Za Zhi. 2011 Apr;47(4):373-7. Zhonghua Yan Ke Za Zhi. 2011. PMID: 21612688 Review. Chinese.
-
Alternative splicing in angiogenesis: the vascular endothelial growth factor paradigm.Cancer Lett. 2007 May 8;249(2):133-42. doi: 10.1016/j.canlet.2006.08.015. Epub 2006 Oct 5. Cancer Lett. 2007. PMID: 17027147 Review.
Cited by
-
The role of VEGF 165b in pathophysiology.Cell Adh Migr. 2012 Nov-Dec;6(6):561-8. doi: 10.4161/cam.22439. Epub 2012 Oct 17. Cell Adh Migr. 2012. PMID: 23076130 Free PMC article. Review.
-
Immunoexpression of CD44, p16 and VEGF in oral cancer.J Oral Maxillofac Pathol. 2024 Apr-Jun;28(2):253-260. doi: 10.4103/jomfp.jomfp_195_23. Epub 2024 Jul 11. J Oral Maxillofac Pathol. 2024. PMID: 39157839 Free PMC article.
-
Development of a naringenin microemulsion as a prospective ophthalmic delivery system for the treatment of corneal neovascularization: in vitro and in vivo evaluation.Drug Deliv. 2022 Dec;29(1):111-127. doi: 10.1080/10717544.2021.2021323. Drug Deliv. 2022. PMID: 34964414 Free PMC article.
-
How VEGF-A and its splice variants affect breast cancer development - clinical implications.Cell Oncol (Dordr). 2022 Apr;45(2):227-239. doi: 10.1007/s13402-022-00665-w. Epub 2022 Mar 18. Cell Oncol (Dordr). 2022. PMID: 35303290 Free PMC article. Review.
-
Splicing Regulators and Their Roles in Cancer Biology and Therapy.Biomed Res Int. 2015;2015:150514. doi: 10.1155/2015/150514. Epub 2015 Jul 26. Biomed Res Int. 2015. PMID: 26273588 Free PMC article. Review.
References
-
- Mueller S, Matthay KK. Neuroblastoma: biology and staging. Curr Oncol Rep. 2009;11:431–438. - PubMed
-
- Pastorino F, Di PD, Loi M, Becherini P, Caffa I, Zorzoli A, et al. Recent advances in targeted anti-vasculature therapy: the neuroblastoma model. Curr Drug Targets. 2009;10:1021–1027. - PubMed
-
- Rossler J, Taylor M, Geoerger B, Farace F, Lagodny J, Peschka-Suss R, et al. Angiogenesis as a target in neuroblastoma. Eur J Cancer. 2008;44:1645–1656. - PubMed
-
- Beierle EA, Strande LF, Chen MK. VEGF upregulates Bcl-2 expression and is associated with decreased apoptosis in neuroblastoma cells. J Pediatr Surg. 2002;37:467–471. - PubMed
-
- Beierle EA, Dai W, Langham MR, Jr., Copeland EM, III, Chen MK. VEGF receptors are differentially expressed by neuroblastoma cells in culture. J Pediatr Surg. 2003;38:514–521. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

