Lysophospholipid micelles sustain the stability and catalytic activity of diacylglycerol kinase in the absence of lipids
- PMID: 20666483
- PMCID: PMC2923640
- DOI: 10.1021/bi100575s
Lysophospholipid micelles sustain the stability and catalytic activity of diacylglycerol kinase in the absence of lipids
Abstract
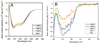
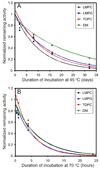
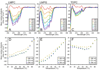
There has been a renewal of interest in interactions of membrane proteins with detergents and lipids, sparked both by recent results that illuminate the structural details of these interactions and also by the realization that some experimental membrane protein structures are distorted by detergent-protein interactions. The integral membrane enzyme diacylglycerol kinase (DAGK) has long been thought to require the presence of lipid as an obligate "cofactor" in order to be catalytically viable in micelles. Here, we report that near-optimal catalytic properties are observed for DAGK in micelles composed of lysomyristoylphosphatidylcholine (LMPC), with significant activity also being observed in micelles composed of lysomyristoylphosphatidylglycerol and tetradecylphosphocholine. All three of these detergents were also sustained high stability of the enzyme. NMR measurements revealed significant differences in DAGK-detergent interactions involving LMPC micelles versus micelles composed of dodecylphosphocholine. These results highlight the fact that some integral membrane proteins can maintain native-like properties in lipid-free detergent micelles and also suggest that C(14)-based detergents may be worthy of more widespread use in studies of membrane proteins.
Figures




Similar articles
-
On choosing a detergent for solution NMR studies of membrane proteins.J Biomol NMR. 1998 May;11(4):381-6. doi: 10.1023/a:1008289624496. J Biomol NMR. 1998. PMID: 9691283
-
Escherichia coli diacylglycerol kinase: a case study in the application of solution NMR methods to an integral membrane protein.Biophys J. 1997 Jun;72(6):2688-701. doi: 10.1016/S0006-3495(97)78912-4. Biophys J. 1997. PMID: 9168044 Free PMC article.
-
Reconstitutive refolding of diacylglycerol kinase, an integral membrane protein.Biochemistry. 1999 Dec 7;38(49):16373-82. doi: 10.1021/bi991292n. Biochemistry. 1999. PMID: 10587463
-
Prokaryotic diacylglycerol kinase and undecaprenol kinase.Annu Rev Biophys. 2012;41:81-101. doi: 10.1146/annurev-biophys-050511-102330. Epub 2011 Dec 20. Annu Rev Biophys. 2012. PMID: 22224599 Free PMC article. Review.
-
NMR solution structure determination of membrane proteins reconstituted in detergent micelles.FEBS Lett. 2003 Nov 27;555(1):144-50. doi: 10.1016/s0014-5793(03)01155-4. FEBS Lett. 2003. PMID: 14630335 Review.
Cited by
-
Structural and functional characterisation of TesA - a novel lysophospholipase A from Pseudomonas aeruginosa.PLoS One. 2013 Jul 18;8(7):e69125. doi: 10.1371/journal.pone.0069125. Print 2013. PLoS One. 2013. PMID: 23874889 Free PMC article.
-
Membrane properties that shape the evolution of membrane enzymes.Curr Opin Struct Biol. 2018 Aug;51:80-91. doi: 10.1016/j.sbi.2018.03.013. Epub 2018 Mar 27. Curr Opin Struct Biol. 2018. PMID: 29597094 Free PMC article. Review.
-
Structure and physiological function of the human KCNQ1 channel voltage sensor intermediate state.Elife. 2020 Feb 25;9:e53901. doi: 10.7554/eLife.53901. Elife. 2020. PMID: 32096762 Free PMC article.
-
Insights into the oligomeric structure of the HIV-1 Vpu protein.J Struct Biol. 2023 Mar;215(1):107943. doi: 10.1016/j.jsb.2023.107943. Epub 2023 Feb 14. J Struct Biol. 2023. PMID: 36796461 Free PMC article.
-
Comparative study of the structure and interaction of the pore helices of the hERG and Kv1.5 potassium channels in model membranes.Eur Biophys J. 2017 Sep;46(6):549-559. doi: 10.1007/s00249-017-1201-2. Epub 2017 Mar 17. Eur Biophys J. 2017. PMID: 28314880
References
-
- Sanders CR, Sonnichsen F. Solution NMR of membrane proteins: practice and challenges. Magn Reson. Chem. 2006;44(Spec No):S24–S40. - PubMed
-
- Czerski L, Sanders CR. Functionality of a membrane protein in bicelles. Anal. Biochem. 2000;284:327–333. - PubMed
-
- Hunte C, Richers S. Lipids and membrane protein structures. Curr. Opin. Struct. Biol. 2008;18:406–411. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

