A multivalent vaccination strategy for the prevention of Old World arenavirus infection in humans
- PMID: 20668086
- PMCID: PMC2937778
- DOI: 10.1128/JVI.00672-10
A multivalent vaccination strategy for the prevention of Old World arenavirus infection in humans
Abstract
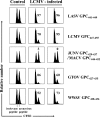
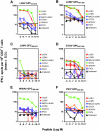
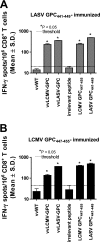
Arenaviruses cause severe human disease ranging from aseptic meningitis following lymphocytic choriomeningitis virus (LCMV) infection to hemorrhagic fever syndromes following infection with Guanarito virus (GTOV), Junin virus (JUNV), Lassa virus (LASV), Machupo virus (MACV), Sabia virus (SABV), or Whitewater Arroyo virus (WWAV). Cellular immunity, chiefly the CD8(+) T-cell response, plays a critical role in providing protective immunity following infection with the Old World arenaviruses LASV and LCMV. In the current study, we evaluated whether HLA class I-restricted epitopes that are cross-reactive among pathogenic arenaviruses could be identified for the purpose of developing an epitope-based vaccination approach that would cross-protect against multiple arenaviruses. We were able to identify a panel of HLA-A*0201-restricted peptides derived from the same region of the glycoprotein precursor (GPC) of LASV (GPC spanning residues 441 to 449 [GPC(441-449)]), LCMV (GPC(447-455)), JUNV (GPC(429-437)), MACV (GPC(444-452)), GTOV (GPC(427-435)), and WWAV (GPC(428-436)) that displayed high-affinity binding to HLA-A*0201 and were recognized by CD8(+) T cells in a cross-reactive manner following LCMV infection or peptide immunization of HLA-A*0201 transgenic mice. Immunization of HLA-A*0201 mice with the Old World peptide LASV GPC(441-449) or LCMV GPC(447-455) induced high-avidity CD8(+) T-cell responses that were able to kill syngeneic target cells pulsed with either LASV GPC(441-449) or LCMV GPC(447-455) in vivo and provided significant protection against viral challenge with LCMV. Through this study, we have demonstrated that HLA class I-restricted, cross-reactive epitopes exist among diverse arenaviruses and that individual epitopes can be utilized as effective vaccine determinants for multiple pathogenic arenaviruses.
Figures

References
-
- Alexander, J., P. Bilsel, M. F. del Guercio, S. Stewart, A. Marinkovic-Petrovic, S. Southwood, C. Crimi, L. Vang, L. Walker, G. Ishioka, V. Chitnis, A. Sette, E. Assarsson, D. Hannaman, J. Botten, and M. J. Newman. 2010. Universal influenza DNA vaccine encoding conserved CD4+ T cell epitopes protects against lethal viral challenge in HLA-DR transgenic mice. Vaccine 28:664-672. - PMC - PubMed
-
- Barton, L. L., S. C. Budd, W. S. Morfitt, C. J. Peters, T. G. Ksiazek, R. F. Schindler, and M. T. Yoshino. 1993. Congenital lymphocytic choriomeningitis virus infection in twins. Pediatr. Infect. Dis. J. 12:942-946. - PubMed
-
- Barton, L. L., M. B. Mets, and C. L. Beauchamp. 2002. Lymphocytic choriomeningitis virus: emerging fetal teratogen. Am. J. Obstet. Gynecol. 187:1715-1716. - PubMed
-
- Bechtel, R. T., K. A. Haught, and M. B. Mets. 1997. Lymphocytic choriomeningitis virus: a new addition to the TORCH evaluation. Arch. Ophthalmol. 115:680-681. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- T32 AI007354/AI/NIAID NIH HHS/United States
- AI065359/AI/NIAID NIH HHS/United States
- R01 AI050840/AI/NIAID NIH HHS/United States
- AI077607/AI/NIAID NIH HHS/United States
- R01 AI074862/AI/NIAID NIH HHS/United States
- R01 AI027028/AI/NIAID NIH HHS/United States
- AI27028/AI/NIAID NIH HHS/United States
- U54 AI065359/AI/NIAID NIH HHS/United States
- AI074862/AI/NIAID NIH HHS/United States
- R21 AI027028/AI/NIAID NIH HHS/United States
- R01 AI077607/AI/NIAID NIH HHS/United States
- P20RR021905/RR/NCRR NIH HHS/United States
- P20 RR021905/RR/NCRR NIH HHS/United States
- R56 AI027028/AI/NIAID NIH HHS/United States
- N01 AI040023/AI/NIAID NIH HHS/United States
- AI50840/AI/NIAID NIH HHS/United States
- F32 AI056827/AI/NIAID NIH HHS/United States
- T32 AI07354/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

