Heat shock protein 90 protects rat mesenchymal stem cells against hypoxia and serum deprivation-induced apoptosis via the PI3K/Akt and ERK1/2 pathways
- PMID: 20669351
- PMCID: PMC2916094
- DOI: 10.1631/jzus.B1001007
Heat shock protein 90 protects rat mesenchymal stem cells against hypoxia and serum deprivation-induced apoptosis via the PI3K/Akt and ERK1/2 pathways
Abstract
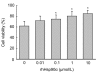
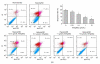
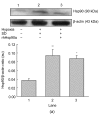
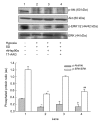
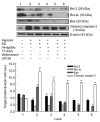
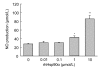
Mesenchymal stem cell (MSC) transplantation has shown a therapeutic potential to repair the ischemic and infracted myocardium, but the effects are limited by the apoptosis and loss of donor cells in host cardiac microenvironment. The aim of this study is to explore the cytoprotection of heat shock protein 90 (Hsp90) against hypoxia and serum deprivation-induced apoptosis and the possible mechanisms in rat MSCs. Cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Apoptosis was assessed by Hoechst 33258 nuclear staining and flow cytometric analysis with annexin V/PI staining. The gene expression of Toll-like receptor-4 (TLR-4) and V-erb-b2 erythroblastic leukemia viral oncogene homolog 2 (ErbB2) was detected by real-time polymerase chain reaction (PCR). The protein levels of cleaved caspase-3, Bcl-2, Bcl-xL, Bax, total-ERK, phospho-ERK, total-Akt, phospho-Akt, and Hsp90 were detected by Western blot. The production of nitric oxide was measured by spectrophotometric assay. Hsp90 improves MSC viability and protects MSCs against apoptosis induced by serum deprivation and hypoxia. The protective role of Hsp90 not only elevates Bcl-2/Bax and Bcl-xL/Bax expression and attenuates cleaved caspase-3 expression via down-regulating membrane TLR-4 and ErbB2 receptors and then activating their downstream PI3K/Akt and ERK1/2 pathways, but also enhances the paracrine effect of MSCs. These findings demonstrated a novel and effective treatment strategy against MSC apoptosis in cell transplantation.
Figures


Similar articles
-
Angiopoietin-like 4 confers resistance to hypoxia/serum deprivation-induced apoptosis through PI3K/Akt and ERK1/2 signaling pathways in mesenchymal stem cells.PLoS One. 2014 Jan 21;9(1):e85808. doi: 10.1371/journal.pone.0085808. eCollection 2014. PLoS One. 2014. Retraction in: PLoS One. 2018 Mar 12;13(3):e0194448. doi: 10.1371/journal.pone.0194448. PMID: 24465718 Free PMC article. Retracted.
-
Lovastatin protects mesenchymal stem cells against hypoxia- and serum deprivation-induced apoptosis by activation of PI3K/Akt and ERK1/2.J Cell Biochem. 2008 Jan 1;103(1):256-69. doi: 10.1002/jcb.21402. J Cell Biochem. 2008. PMID: 17497701
-
[Lovastatin protects mesenchymal stem cells against hypoxia and serum deprivation-induced apoptosis through activation of PI3K/Akt and ERK1/2 signaling pathways].Zhonghua Xin Xue Guan Bing Za Zhi. 2008 Aug;36(8):685-90. Zhonghua Xin Xue Guan Bing Za Zhi. 2008. PMID: 19100107 Chinese.
-
Lysophosphatidic acid protects mesenchymal stem cells against hypoxia and serum deprivation-induced apoptosis.Stem Cells. 2008 Jan;26(1):135-45. doi: 10.1634/stemcells.2007-0098. Epub 2007 Oct 11. Stem Cells. 2008. PMID: 17932426
-
How to enhance MSCs therapeutic properties? An insight on potentiation methods.Stem Cell Res Ther. 2024 Sep 27;15(1):331. doi: 10.1186/s13287-024-03935-6. Stem Cell Res Ther. 2024. PMID: 39334487 Free PMC article. Review.
Cited by
-
Effects of Hypoxia on RNA Cargo in Extracellular Vesicles from Human Adipose-Derived Stromal/Stem Cells.Int J Mol Sci. 2022 Jul 2;23(13):7384. doi: 10.3390/ijms23137384. Int J Mol Sci. 2022. PMID: 35806391 Free PMC article.
-
Heat shock pretreatment improves mesenchymal stem cell viability by heat shock proteins and autophagy to prevent cisplatin-induced granulosa cell apoptosis.Stem Cell Res Ther. 2019 Nov 26;10(1):348. doi: 10.1186/s13287-019-1425-4. Stem Cell Res Ther. 2019. Retraction in: Stem Cell Res Ther. 2025 Jul 1;16(1):323. doi: 10.1186/s13287-025-04486-0. PMID: 31771642 Free PMC article. Retracted.
-
Localisation Microscopy of Breast Epithelial ErbB-2 Receptors and Gap Junctions: Trafficking after γ-Irradiation, Neuregulin-1β, and Trastuzumab Application.Int J Mol Sci. 2017 Feb 9;18(2):362. doi: 10.3390/ijms18020362. Int J Mol Sci. 2017. PMID: 28208769 Free PMC article.
-
State of the Art Review of Cell Therapy in the Treatment of Lung Disease, and the Potential for Aerosol Delivery.Int J Mol Sci. 2020 Sep 3;21(17):6435. doi: 10.3390/ijms21176435. Int J Mol Sci. 2020. PMID: 32899381 Free PMC article. Review.
-
A revealing review of mesenchymal stem cells therapy, clinical perspectives and Modification strategies.Stem Cell Investig. 2019 Sep 25;6:34. doi: 10.21037/sci.2019.08.11. eCollection 2019. Stem Cell Investig. 2019. PMID: 31620481 Free PMC article. Review.
References
-
- Chen TL, Wang JA, Shi H, Gui C, Luo RH, Xie XJ, Xiang MX, Zhang X, Cao J. Cyclosporin A pre-incubation attenuates hypoxia/reoxygenation-induced apoptosis in mesenchymal stem cells. Scandinavian Journal of Clinical & Laboratory Investigation. 2008;68(7):585–593. doi: 10.1080/00365510801918761. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

