Functionalized congener approach to muscarinic antagonists: analogues of pirenzepine
- PMID: 2066986
- PMCID: PMC3469255
- DOI: 10.1021/jm00111a032
Functionalized congener approach to muscarinic antagonists: analogues of pirenzepine
Abstract
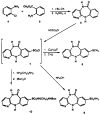
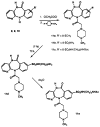
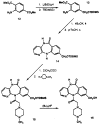
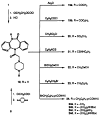
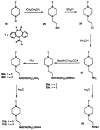
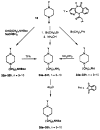
The M1-selective muscarinic receptor antagonist pirenzepine 6H-pyrido[2,3-b][1,4]benzodiazepin-6-one) was derivatized to explore points of attachment of functionalized side chains for the synthesis of receptor probes and ligands for affinity chromatography. The analogues prepared were evaluated in competitive binding assays versus [3H]-N-methylscopolamine at four muscarinic receptor subtypes (m1AChR-m4AChR) in membranes from rat heart tissue and transfected A9L cells. 9-(Hydroxymethyl)pirenzepine, 8-(methylthio)pirenzepine, and a series of 8-aminosulfonyl derivatives were synthesized. Several 5-substituted analogues of pirenzepine also were prepared. An alternate series of analogues substituted on the 4-position of the piperazine ring was prepared by reaction of 4-desmethylpirenzepine with various electrophiles. An N-chloroethyl analogue of pirenzepine was shown to form a reactive aziridine species in aqueous buffer yet failed to affinity label muscarinic receptors. Within a series of aminoalkyl analogues, the affinity increased as the length of the alkyl chain increased. Shorter chain analogues were generally much less potent than pirenzepine, and longer analogues (7-10 carbons) were roughly as potent as pirenzepine at m1 receptors, but were nonselective. Depending on the methylene chain length, acylation or alkyl substitution of the terminal amine also influenced the affinity at muscarinic receptors.
Figures

Similar articles
-
Molecular probes for muscarinic receptors: derivatives of the M1-antagonist telenzepine.Bioconjug Chem. 1992 May-Jun;3(3):234-40. doi: 10.1021/bc00015a006. Bioconjug Chem. 1992. PMID: 1520727 Free PMC article.
-
Regional distribution of M1, M2 and non-M1, non-M2 subtypes of muscarinic binding sites in rat brain.J Pharmacol Exp Ther. 1990 Dec;255(3):1148-57. J Pharmacol Exp Ther. 1990. PMID: 2262898
-
N-[3H]methylscopolamine labeling of non-M1, non-M2 muscarinic receptor binding sites in rat brain.J Pharmacol Exp Ther. 1991 Mar;256(3):1173-81. J Pharmacol Exp Ther. 1991. PMID: 2005579
-
Multiple binding affinities of N-methylscopolamine to brain muscarinic acetylcholine receptors: differentiation from M1 and M2 receptor subtypes.J Pharmacol Exp Ther. 1986 Aug;238(2):554-63. J Pharmacol Exp Ther. 1986. PMID: 3755473
-
Molecular probes for muscarinic receptors: functionalized congeners of selective muscarinic antagonists.Life Sci. 1995;56(11-12):823-30. doi: 10.1016/0024-3205(95)00016-y. Life Sci. 1995. PMID: 10188781 Free PMC article. Review.
Cited by
-
Molecular probes for muscarinic receptors: derivatives of the M1-antagonist telenzepine.Bioconjug Chem. 1992 May-Jun;3(3):234-40. doi: 10.1021/bc00015a006. Bioconjug Chem. 1992. PMID: 1520727 Free PMC article.
-
Unexpected scaffold rearrangement product of pirenzepine found in commercial samples.Sci Rep. 2021 Dec 3;11(1):23397. doi: 10.1038/s41598-021-02732-y. Sci Rep. 2021. PMID: 34862437 Free PMC article.
-
Functionalized congener approach to the design of ligands for G protein-coupled receptors (GPCRs).Bioconjug Chem. 2009 Oct 21;20(10):1816-35. doi: 10.1021/bc9000596. Epub 2009 Apr 30. Bioconjug Chem. 2009. PMID: 19405524 Free PMC article. Review.
-
High affinity acylating antagonists for muscarinic receptors.Life Sci. 1992;51(5):345-51. doi: 10.1016/0024-3205(92)90586-e. Life Sci. 1992. PMID: 1625525 Free PMC article.
-
Amphiphilic pyridinium salts block TNF alpha/NF kappa B signaling and constitutive hypersecretion of interleukin-8 (IL-8) from cystic fibrosis lung epithelial cells.Biochem Pharmacol. 2005 Aug 1;70(3):381-93. doi: 10.1016/j.bcp.2005.05.002. Biochem Pharmacol. 2005. PMID: 15963954 Free PMC article.
References
-
- Frotscher M, Misgeld U, editors. Central Cholinergic Synaptic Transmission; Birkhauser Verlag; Basel: 1989.
-
- Kromer W, Gönne S. Int J Exp Clin Pharmacol. 1988;37(suppl 1):48. - PubMed
-
- Goyal R. New Engl J Med. 1989;321:1022. - PubMed
-
- Maclagan J, Barnes P. Trends Phar Sci. 1989:88–92. Suppl. “Subtypes of Muscarinic Receptors IV”. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
