Allogeneic T cells impair engraftment and hematopoiesis after stem cell transplantation
- PMID: 20679222
- PMCID: PMC2930440
- DOI: 10.1073/pnas.1009220107
Allogeneic T cells impair engraftment and hematopoiesis after stem cell transplantation
Abstract
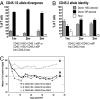
Because of the perception that depleting hematopoietic grafts of T cells will result in poorer immune recovery and in increased risk of graft rejection, pure hematopoietic stem cells (HSC), which avoid the potentially lethal complication of graft-versus-host disease (GVHD), have not been used for allogeneic hematopoietic cell transplantation (HCT) in humans. Ideal grafts should contain HSC plus mature cells that confer only the benefits of protection from pathogens and suppression of malignancies. This goal requires better understanding of the effects of each blood cell type and its interactions during engraftment and immune regeneration. Here, we studied hematopoietic reconstitution post-HCT, comparing grafts of purified HSC with grafts supplemented with T cells in a minor histocompatibility antigen (mHA)-mismatched mouse model. Cell counts, composition, and chimerism of blood and lymphoid organs were evaluated and followed intensively through the first month, and then subsequently for up to 1 yr. Throughout this period, recipients of pure HSC demonstrated superior total cell recovery and lymphoid reconstitution compared with recipients of T cell-containing grafts. In the latter, rapid expansion of T cells occurred, and suppression of hematopoiesis derived from donor HSC was observed. Our findings demonstrate that even early post-HCT, T cells retard donor HSC engraftment and immune recovery. These observations contradict the postulation that mature donor T cells provide important transient immunity and facilitate HSC engraftment.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Allogeneic bone marrow grafts with high levels of CD4(+) CD25(+) FoxP3(+) T cells can lead to engraftment failure.Cytometry A. 2012 Jun;81(6):476-88. doi: 10.1002/cyto.a.22061. Epub 2012 Apr 20. Cytometry A. 2012. PMID: 22522779
-
Donor hematopoiesis in mice following total lymphoid irradiation requires host T-regulatory cells for durable engraftment.Blood. 2014 May 1;123(18):2882-92. doi: 10.1182/blood-2013-10-530212. Epub 2014 Mar 3. Blood. 2014. PMID: 24591203 Free PMC article.
-
Purified hematopoietic stem cell grafts induce tolerance to alloantigens and can mediate positive and negative T cell selection.Proc Natl Acad Sci U S A. 2000 Aug 15;97(17):9555-60. doi: 10.1073/pnas.170279297. Proc Natl Acad Sci U S A. 2000. PMID: 10920206 Free PMC article.
-
Immune reconstitution and tolerance after allogeneic hematopoietic stem cell transplantation.Hematology. 2003 Feb;8(1):19-26. doi: 10.1080/1024533031000072045. Hematology. 2003. PMID: 12623423 Review.
-
Protective conditioning against GVHD and graft rejection after combined organ and hematopoietic cell transplantation.Blood Cells Mol Dis. 2008 Jan-Feb;40(1):48-54. doi: 10.1016/j.bcmd.2007.06.019. Epub 2007 Sep 10. Blood Cells Mol Dis. 2008. PMID: 17827036 Review.
Cited by
-
Competition between T cells maintains clonal dominance during memory inflation induced by MCMV.Eur J Immunol. 2013 May;43(5):1252-63. doi: 10.1002/eji.201242940. Epub 2013 Mar 20. Eur J Immunol. 2013. PMID: 23404526 Free PMC article.
-
Blood Stem Cell Activity Is Arrested by Th1-Mediated Injury Preventing Engraftment following Nonmyeloablative Conditioning.J Immunol. 2016 Nov 15;197(10):4151-4162. doi: 10.4049/jimmunol.1500715. Epub 2016 Oct 10. J Immunol. 2016. PMID: 27815446 Free PMC article.
-
Spatio-Temporal Bone Remodeling after Hematopoietic Stem Cell Transplantation.Int J Mol Sci. 2020 Dec 29;22(1):267. doi: 10.3390/ijms22010267. Int J Mol Sci. 2020. PMID: 33383915 Free PMC article.
-
Deletion of AMPK minimizes graft-versus-host disease through an early impact on effector donor T cells.JCI Insight. 2021 Jul 22;6(14):e143811. doi: 10.1172/jci.insight.143811. JCI Insight. 2021. PMID: 34291733 Free PMC article.
-
IL-22-dependent dysbiosis and mononuclear phagocyte depletion contribute to steroid-resistant gut graft-versus-host disease in mice.Nat Commun. 2021 Feb 5;12(1):805. doi: 10.1038/s41467-021-21133-3. Nat Commun. 2021. PMID: 33547295 Free PMC article.
References
-
- Horowitz M. Uses and growth of hematopoietic cell transplantation. In: Appelbaum FR, Forman SJ, Negrin RS, Blume KG, editors. Thomas’ Hematopoietic Cell Transplantation. 4th Ed. Oxford, UK: Wiley-Blackwell; 2009. pp. 15–21.
-
- Scandling JD, et al. Tolerance and chimerism after renal and hematopoietic-cell transplantation. N Engl J Med. 2008;358:362–368. - PubMed
-
- Martin PJ, et al. Graft failure in patients receiving T cell-depleted HLA-identical allogeneic marrow transplants. Bone Marrow Transplant. 1988;3:445–456. - PubMed
-
- Marmont AM, et al. T-cell depletion of HLA-identical transplants in leukemia. Blood. 1991;78:2120–2130. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

