Tumor-supportive and osteoclastogenic changes induced by breast cancer-derived factors are reversed by inhibition of {gamma}-secretase
- PMID: 20679341
- PMCID: PMC2951217
- DOI: 10.1074/jbc.M110.114496
Tumor-supportive and osteoclastogenic changes induced by breast cancer-derived factors are reversed by inhibition of {gamma}-secretase
Abstract
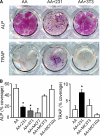
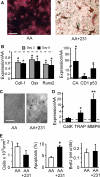
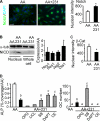
During breast cancer metastasis to bone, tumor cells home to bone marrow, likely targeting the stem cell niche, and stimulate osteoclasts, which mediate osteolysis required for tumor expansion. Although osteoblasts contribute to the regulation of the hematopoietic stem cell niche and control osteoclastogenesis through production of proresorptive cytokine RANKL (receptor activator of NF-κB ligand), their role in cancer metastases to bone is not fully understood. C57BL/6J mouse bone marrow cells were treated for 3-12 days with ascorbic acid (50 μg/ml) in the presence or absence of 10% medium conditioned by breast carcinoma cells MDA-MB-231, 4T1, or MCF7. Treatment with cancer-derived factors resulted in a sustained 40-60% decrease in osteoblast differentiation markers, compared with treatment with ascorbic acid alone, and induced an osteoclastogenic change in the RANKL/osteoprotegerin ratio. Importantly, exposure of bone cells to breast cancer-derived factors stimulated the subsequent attachment of cancer cells to immature osteoblasts. Inhibition of γ-secretase using pharmacological inhibitors DAPT and Compound E completely reversed cancer-induced osteoclastogenesis as well as cancer-induced enhancement of cancer cell attachment, identifying γ-secretase activity as a key mediator of these effects. Thus, we have uncovered osteoblasts as critical intermediary of premetastatic signaling by breast cancer cells and pinpointed γ-secretase as a robust target for developing therapeutics potentially capable of reducing both homing and progression of cancer metastases to bone.
Figures
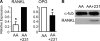
Similar articles
-
Expression of osteoprotegerin and RANK ligand in breast cancer bone metastasis.J Korean Med Sci. 2003 Aug;18(4):541-6. doi: 10.3346/jkms.2003.18.4.541. J Korean Med Sci. 2003. PMID: 12923331 Free PMC article.
-
Pharmacological Inhibition of the Skeletal IKKβ Reduces Breast Cancer-Induced Osteolysis.Calcif Tissue Int. 2018 Aug;103(2):206-216. doi: 10.1007/s00223-018-0406-4. Epub 2018 Feb 17. Calcif Tissue Int. 2018. PMID: 29455416 Free PMC article.
-
DU145 human prostate cancer cells express functional receptor activator of NFkappaB: new insights in the prostate cancer bone metastasis process.Bone. 2007 Apr;40(4):981-90. doi: 10.1016/j.bone.2006.11.006. Epub 2006 Dec 28. Bone. 2007. PMID: 17196895
-
Breast cancer metastasis to the bone: mechanisms of bone loss.Breast Cancer Res. 2010;12(6):215. doi: 10.1186/bcr2781. Epub 2010 Dec 16. Breast Cancer Res. 2010. PMID: 21176175 Free PMC article. Review.
-
Osteoclast differentiation by RANKL and OPG signaling pathways.J Bone Miner Metab. 2021 Jan;39(1):19-26. doi: 10.1007/s00774-020-01162-6. Epub 2020 Oct 20. J Bone Miner Metab. 2021. PMID: 33079279 Review.
Cited by
-
Breast cancer at bone metastatic sites: recent discoveries and treatment targets.J Cell Commun Signal. 2011 Jun;5(2):85-99. doi: 10.1007/s12079-011-0117-3. Epub 2011 Jan 19. J Cell Commun Signal. 2011. PMID: 21484191 Free PMC article.
-
Role of Notch and its oncogenic signaling crosstalk in breast cancer.Biochim Biophys Acta. 2011 Apr;1815(2):197-213. doi: 10.1016/j.bbcan.2010.12.002. Epub 2010 Dec 28. Biochim Biophys Acta. 2011. PMID: 21193018 Free PMC article. Review.
-
Walker 256/B malignant breast cancer cells improve femur angioarchitecture and disrupt hematological parameters in a rat model of tumor osteolysis.Tumour Biol. 2014 Apr;35(4):3663-70. doi: 10.1007/s13277-013-1485-5. Epub 2013 Dec 8. Tumour Biol. 2014. PMID: 24318993
-
Three-Dimensional Mechanical Loading Modulates the Osteogenic Response of Mesenchymal Stem Cells to Tumor-Derived Soluble Signals.Tissue Eng Part A. 2016 Aug;22(15-16):1006-15. doi: 10.1089/ten.TEA.2016.0153. Epub 2016 Aug 1. Tissue Eng Part A. 2016. PMID: 27401765 Free PMC article.
-
Pathway and network analysis of genes related to osteoporosis.Mol Med Rep. 2019 Aug;20(2):985-994. doi: 10.3892/mmr.2019.10353. Epub 2019 Jun 6. Mol Med Rep. 2019. PMID: 31173222 Free PMC article.
References
-
- Coleman R. E. (2006) Clin. Cancer Res. 12, 6243S–6249S - PubMed
-
- Kim C. J., Bland K. I., Yeatman T. J. (2004) The Breast Comprehensive Management of Benign and Malignant Disorders, W. B. Saunders, Philadelphia, PA
-
- Hattner R., Epker B. N., Frost H. M. (1965) Nature 206, 489–490 - PubMed
-
- Stenbeck G. (2002) Semin. Cell Dev. Biol. 13, 285–292 - PubMed
-
- Deng Z. L., Sharff K. A., Tang N., Song W. X., Luo J., Luo X., Chen J., Bennett E., Reid R., Manning D., Xue A., Montag A. G., Luu H. H., Haydon R. C., He T. C. (2008) Front. Biosci. 13, 2001–2021 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

