Overcoming the heme paradox: heme toxicity and tolerance in bacterial pathogens
- PMID: 20679437
- PMCID: PMC2981329
- DOI: 10.1128/IAI.00613-10
Overcoming the heme paradox: heme toxicity and tolerance in bacterial pathogens
Abstract
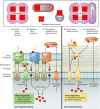
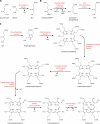
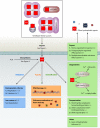
Virtually all bacterial pathogens require iron to infect vertebrates. The most abundant source of iron within vertebrates is in the form of heme as a cofactor of hemoproteins. Many bacterial pathogens have elegant systems dedicated to the acquisition of heme from host hemoproteins. Once internalized, heme is either degraded to release free iron or used intact as a cofactor in catalases, cytochromes, and other bacterial hemoproteins. Paradoxically, the high redox potential of heme makes it a liability, as heme is toxic at high concentrations. Although a variety of mechanisms have been proposed to explain heme toxicity, the mechanisms by which heme kills bacteria are not well understood. Nonetheless, bacteria employ various strategies to protect against and eliminate heme toxicity. Factors involved in heme acquisition and detoxification have been found to contribute to virulence, underscoring the physiological relevance of heme stress during pathogenesis. Herein we describe the current understanding of the mechanisms of heme toxicity and how bacterial pathogens overcome the heme paradox during infection.
Figures
References
-
- Aft, R. L., and G. C. Mueller. 1983. Hemin-mediated DNA strand scission. J. Biol. Chem. 258:12069-12072. - PubMed
-
- Aft, R. L., and G. C. Mueller. 1984. Hemin-mediated oxidative degradation of proteins. J. Biol. Chem. 259:301-305. - PubMed
-
- Arnoux, P., R. Haser, N. Izadi-Pruneyre, A. Lecroisey, and M. Czjzek. 2000. Functional aspects of the heme bound hemophore HasA by structural analysis of various crystal forms. Proteins 41:202-210. - PubMed
-
- Bibb, L. A., N. D. King, C. A. Kunkle, and M. P. Schmitt. 2005. Analysis of a heme-dependent signal transduction system in Corynebacterium diphtheriae: deletion of the chrAS genes results in heme sensitivity and diminished heme-dependent activation of the hmuO promoter. Infect. Immun. 73:7406-7412. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

