Requirement of JIP1-mediated c-Jun N-terminal kinase activation for obesity-induced insulin resistance
- PMID: 20679483
- PMCID: PMC2950523
- DOI: 10.1128/MCB.00585-10
Requirement of JIP1-mediated c-Jun N-terminal kinase activation for obesity-induced insulin resistance
Abstract
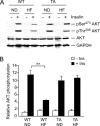
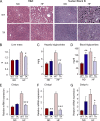
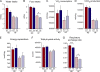
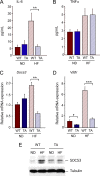
The c-Jun NH(2)-terminal kinase (JNK) interacting protein 1 (JIP1) has been proposed to act as a scaffold protein that mediates JNK activation. However, recent studies have implicated JIP1 in multiple biochemical processes. Physiological roles of JIP1 that are related to the JNK scaffold function of JIP1 are therefore unclear. To test the role of JIP1 in JNK activation, we created mice with a germ line point mutation in the Jip1 gene (Thr(103) replaced with Ala) that selectively blocks JIP1-mediated JNK activation. These mutant mice exhibit a severe defect in JNK activation caused by feeding of a high-fat diet. The loss of JIP1-mediated JNK activation protected the mutant mice against obesity-induced insulin resistance. We conclude that JIP1-mediated JNK activation plays a critical role in metabolic stress regulation of the JNK signaling pathway.
Figures
Similar articles
-
A Protein Scaffold Coordinates SRC-Mediated JNK Activation in Response to Metabolic Stress.Cell Rep. 2017 Sep 19;20(12):2775-2783. doi: 10.1016/j.celrep.2017.08.025. Cell Rep. 2017. PMID: 28930674 Free PMC article.
-
JIP1-Mediated JNK Activation Negatively Regulates Synaptic Plasticity and Spatial Memory.J Neurosci. 2018 Apr 11;38(15):3708-3728. doi: 10.1523/JNEUROSCI.1913-17.2018. Epub 2018 Mar 14. J Neurosci. 2018. PMID: 29540552 Free PMC article.
-
Signal transduction cross talk mediated by Jun N-terminal kinase-interacting protein and insulin receptor substrate scaffold protein complexes.Mol Cell Biol. 2009 Sep;29(17):4831-40. doi: 10.1128/MCB.00155-09. Epub 2009 Jun 29. Mol Cell Biol. 2009. PMID: 19564410 Free PMC article.
-
Role of c-Jun N-terminal Kinase (JNK) in Obesity and Type 2 Diabetes.Cells. 2020 Mar 13;9(3):706. doi: 10.3390/cells9030706. Cells. 2020. PMID: 32183037 Free PMC article. Review.
-
Human Protein Kinases and Obesity.Adv Exp Med Biol. 2017;960:111-134. doi: 10.1007/978-3-319-48382-5_5. Adv Exp Med Biol. 2017. PMID: 28585197 Review.
Cited by
-
Structural mechanisms of allostery and autoinhibition in JNK family kinases.Structure. 2012 Dec 5;20(12):2174-84. doi: 10.1016/j.str.2012.09.021. Epub 2012 Nov 8. Structure. 2012. PMID: 23142346 Free PMC article.
-
The JAX Synteny Browser for mouse-human comparative genomics.Mamm Genome. 2019 Dec;30(11-12):353-361. doi: 10.1007/s00335-019-09821-4. Epub 2019 Nov 27. Mamm Genome. 2019. PMID: 31776723 Free PMC article.
-
A Protein Scaffold Coordinates SRC-Mediated JNK Activation in Response to Metabolic Stress.Cell Rep. 2017 Sep 19;20(12):2775-2783. doi: 10.1016/j.celrep.2017.08.025. Cell Rep. 2017. PMID: 28930674 Free PMC article.
-
Cell Signaling and Stress Responses.Cold Spring Harb Perspect Biol. 2016 Oct 3;8(10):a006072. doi: 10.1101/cshperspect.a006072. Cold Spring Harb Perspect Biol. 2016. PMID: 27698029 Free PMC article. Review.
-
The Regulation of JNK Signaling Pathways in Cell Death through the Interplay with Mitochondrial SAB and Upstream Post-Translational Effects.Int J Mol Sci. 2018 Nov 20;19(11):3657. doi: 10.3390/ijms19113657. Int J Mol Sci. 2018. PMID: 30463289 Free PMC article. Review.
References
-
- Belgardt, B. F., J. Mauer, F. T. Wunderlich, M. B. Ernst, M. Pal, G. Spohn, H. S. Bronneke, S. Brodesser, B. Hampel, A. C. Schauss, and J. C. Bruning. 2010. Hypothalamic and pituitary c-Jun N-terminal kinase 1 signaling coordinately regulates glucose metabolism. Proc. Natl. Acad. Sci. U. S. A. 107:6028-6033. - PMC - PubMed
-
- Emanuelli, B., P. Peraldi, C. Filloux, C. Chavey, K. Freidinger, D. J. Hilton, G. S. Hotamisligil, and E. Van Obberghen. 2001. SOCS-3 inhibits insulin signaling and is up-regulated in response to tumor necrosis factor-alpha in the adipose tissue of obese mice. J. Biol. Chem. 276:47944-47949. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
