Bioavailability and population pharmacokinetics of voriconazole in lung transplant recipients
- PMID: 20679503
- PMCID: PMC2944566
- DOI: 10.1128/AAC.00504-10
Bioavailability and population pharmacokinetics of voriconazole in lung transplant recipients
Abstract
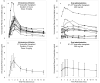
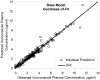
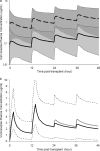
This study was undertaken to characterize the pharmacokinetics and bioavailability of voriconazole in adult lung transplant patients during the early postoperative period, identify factors significantly associated with various pharmacokinetic parameters, and make recommendations for adequate dosing regimens. Thirteen lung transplant patients received two intravenous infusions (6 mg/kg, twice daily [b.i.d.]) immediately posttransplant followed by oral doses (200 mg, b.i.d.) for prophylaxis. Blood samples (9/interval) were collected during one intravenous and one oral dosing interval from each patient. Voriconazole plasma concentrations were measured by high-pressure liquid chromatography (HPLC). NONMEM was used to develop pharmacokinetic models, evaluate covariate relationships, and perform Monte Carlo simulations. There was a good correlation (R(2) = 0.98) between the area under the concentration-time curve specific for the dose evaluated (AUC(0-∞)) and trough concentrations. A two-compartment model adequately described the data. Population estimates of bioavailability, clearance, V(c), and V(p) were 45.9%, 3.45 liters/h, 54.7 liters, and 143 liters. Patients with cystic fibrosis (CF) exhibited a significantly lower bioavailability (23.7%, n = 3) than non-CF patients (63.3%, n = 10). Bioavailability increased with postoperative time and reached steady levels in about 1 week. V(p) increased with body weight. Bioavailability of voriconazole is substantially lower in lung transplant patients than non-transplant subjects but significantly increases with postoperative time. CF patients exhibit significantly lower bioavailability and exposure of voriconazole and therefore need higher doses. Intravenous administration of voriconazole during the first postoperative day followed by oral doses of 200 mg or 400 mg appeared to be the optimal dosing regimen. However, voriconazole levels should be monitored, and the dose should be individualized based on trough concentrations as a good measure of drug exposure.
Figures


References
-
- Berge, M., R. Guillemain, V. Boussaud, M. H. Pham, P. Chevalier, A. Batisse, C. Amrein, E. Dannaoui, M. A. Loriot, A. Lillo-Le Louet, and E. M. Billaud. 2009. Voriconazole pharmacokinetic variability in cystic fibrosis lung transplant patients. Transpl. Infect. Dis. 11:211-219. - PubMed
-
- Bowers, L. D., and D. M. Canafax. 1984. Cyclosporine: experience with therapeutic monitoring. Ther. Drug Monit. 6:142-147. - PubMed
-
- Boyd, A. E., S. Modi, S. J. Howard, C. B. Moore, B. G. Keevil, and D. W. Denning. 2004. Adverse reactions to voriconazole. Clin. Infect. Dis. 39:1241-1244. - PubMed
-
- Christenson, J. T., M. Schmuziger, J. Maurice, F. Simonet, and V. Velebit. 1994. Postoperative visceral hypotension the common cause for gastrointestinal complications after cardiac surgery. Thorac. Cardiovasc. Surg. 42:152-157. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

