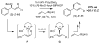Pd-catalyzed enantioselective allyl-allyl cross-coupling
- PMID: 20681700
- PMCID: PMC2925184
- DOI: 10.1021/ja105161f
Pd-catalyzed enantioselective allyl-allyl cross-coupling
Abstract
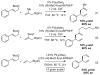
The Pd-catalyzed cross-coupling of allylic carbonates and allylB(pin) is described. The regioselectivity of this reaction is sensitive to the bite angle of the ligand, with small-bite-angle ligands favoring the branched substitution product. This mode of regioselection is consistent with a reaction that operates by a 3,3' reductive elimination reaction. In the presence of appropriate chiral ligands, this reaction is rendered enantioselective and applies to both aromatic and aliphatic allylic carbonates.
Figures
Similar articles
-
Highly selective palladium catalyzed kinetic resolution and enantioselective substitution of racemic allylic carbonates with sulfur nucleophiles: asymmetric synthesis of allylic sulfides, allylic sulfones, and allylic alcohols.Chemistry. 2003 Sep 5;9(17):4202-21. doi: 10.1002/chem.200204657. Chemistry. 2003. PMID: 12953206
-
Direct, intermolecular, enantioselective, iridium-catalyzed allylation of carbamates to form carbamate-protected, branched allylic amines.Org Lett. 2009 Jul 2;11(13):2944-7. doi: 10.1021/ol901151u. Org Lett. 2009. PMID: 19552468 Free PMC article.
-
A Cu/Pd Cooperative Catalysis for Enantioselective Allylboration of Alkenes.J Am Chem Soc. 2015 Nov 4;137(43):13760-3. doi: 10.1021/jacs.5b09146. Epub 2015 Oct 22. J Am Chem Soc. 2015. PMID: 26458555
-
Ligand controlled highly regio- and enantioselective synthesis of alpha-acyloxyketones by palladium-catalyzed allylic alkylation of 1,2-enediol carbonates.J Am Chem Soc. 2008 Sep 10;130(36):11852-3. doi: 10.1021/ja8038954. Epub 2008 Aug 19. J Am Chem Soc. 2008. PMID: 18710230 Free PMC article.
-
Sequential catalytic isomerization and allylic substitution. Conversion of racemic branched allylic carbonates to enantioenriched allylic substitution products.J Am Chem Soc. 2006 Sep 13;128(36):11770-1. doi: 10.1021/ja0644273. J Am Chem Soc. 2006. PMID: 16953612
Cited by
-
Rh-catalyzed regio-switchable cross-coupling of gem-difluorinated cyclopropanes with allylboronates to structurally diverse fluorinated dienes.Chem Sci. 2022 Oct 5;13(42):12419-12425. doi: 10.1039/d2sc04118a. eCollection 2022 Nov 2. Chem Sci. 2022. PMID: 36382270 Free PMC article.
-
Branched/linear selectivity in palladium-catalyzed allyl-allyl cross-couplings: The role of ligands.Tetrahedron. 2015 Sep 16;71(37):6409-6413. doi: 10.1016/j.tet.2015.05.015. Tetrahedron. 2015. PMID: 26279591 Free PMC article.
-
Development and Investigation of a Site Selective Palladium-Catalyzed 1,4-Difunctionalization of Isoprene using Pyridine-Oxazoline Ligands.Chem Sci. 2015 Feb;6(2):1355-1361. doi: 10.1039/C4SC03074E. Chem Sci. 2015. PMID: 25705367 Free PMC article.
-
Catalytic stereospecific allyl-allyl cross-coupling of internal allyl electrophiles with AllylB(pin).Org Lett. 2014 Apr 18;16(8):2096-9. doi: 10.1021/ol500456s. Epub 2014 Apr 4. Org Lett. 2014. PMID: 24702203 Free PMC article.
-
Migratory decarboxylative coupling of coumarins: synthetic and mechanistic aspects.Angew Chem Int Ed Engl. 2011 May 23;50(22):5157-61. doi: 10.1002/anie.201100765. Epub 2011 Apr 19. Angew Chem Int Ed Engl. 2011. PMID: 21506221 Free PMC article. No abstract available.
References
-
- de Meijere A, Diederich F, editors. Metal-Catalyzed Cross-Coupling Reactions. Wiley-VCH; New York: 2004.
-
-
For cross coupling of aryl electrophiles with prochiral allyl metal reagents, see: Sn: Echavarren AM, Stille JK. J Am Chem Soc. 1987;109:5478.Obora Y, Tsuji Y, Kobayashi M, Kawamura T. J Org Chem. 1995;60:4647.Si: Hatanaka Y, Ebina Y, Hiyama T. J Am Chem Soc. 1991;113:7075.Hatanaka Y, Goda K, Hiyama T. Tetrahedron Lett. 1994;35:1279.Hatanaka Y, Goda K, Hiyama T. Tetrahedron Lett. 1994;35:6511.Boron: Kalinin VN, Denisov FS, Bubnov YN. Mendeleev, Commun. 1996. p. 206.Yamamoto Y, Takada S, Miyaura N. Chem Lett. 2006;35:704.
-
-
- Yamamoto Y, Takada S, Miyaura N. Chem Lett. 2006;35:1368.
- Yamamoto Y, Takada S, Miyaura N, Iyama T, Tachikawa H. Organometallics. 2009;28:152.
-
-
Allylstannanes: Trost BM, Keinan E. Tetrahedron Lett. 1980;21:2595.Godschalx J, Stille JK. Tetrahedron Lett. 1980;21:2599.Keinan E, Peretz M. J Org Chem. 1983;48:5302.Trost BM, Pietrusiewicz KM. Tetrahedron Lett. 1985;26:4039.Goliaszewski A, Schwartz J. Tetrahedron. 1985;41:5779.Keinan E, Bosch E. J Org Chem. 1986;51:4006.Cuerva JM, Gómez-Bengoa E, Méndez M, Echavarren AM. J Org Chem. 1997;62:7540.van Heerden FR, Huyser JJ, Williams DBG, Holzapfel CW. Tetrahedron Lett. 1998;39:5281.Nakamura H, Bao M, Yamamoto Y. Angew Chem Int Ed. 2001;40:3208.Méndez M, Cuerva JM, Gómez-Bengoa E, Cárdenas DJ, Echavarren AM. Chem Eur J. 2002;8:3620.While this work was in progress, the cross-coupling of allyl boronates and allyl carbonates appeared: Flegeau EF, Schneider U, Kobayashi S. Chem Eur J. 2009;15:12247.
-
-
- Keinan E, Kumar S, Dangur V, Vaya J. J Am Chem Soc. 1994;116:11151.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources