Immunolocalization of estrogen-producing and metabolizing enzymes in benign breast disease: comparison with normal breast and breast carcinoma
- PMID: 20682005
- PMCID: PMC11159500
- DOI: 10.1111/j.1349-7006.2010.01673.x
Immunolocalization of estrogen-producing and metabolizing enzymes in benign breast disease: comparison with normal breast and breast carcinoma
Abstract
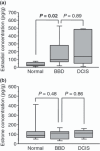
It is well known that estrogens play important roles in the cell proliferation of breast carcinoma. Benign breast disease (BBD) contains a wide spectrum of diseases, and some are considered an important risk factor for subsequent breast carcinoma development. However, the significance of estrogens in BBD has remained largely unknown. Therefore, in this study, we examined tissue concentrations of estrogens and immunolocalization of estrogen-producing/metabolizing enzymes in BBD, and compared these findings with those in the normal breast and ductal carcinoma in situ (DCIS). Tissue concentration of estradiol in BBD (n = 9) was significantly (3.4-fold) higher than normal breast (n = 9) and nearly the same (0.7-fold) as in DCIS (n = 9). Immunoreactivity of estrogen sulfotransferase in BBD was significantly lower (n = 82) than normal breast (n = 28) but was not significantly different from DCIS (n = 28). Aromatase and steroid sulfatase immunoreactivities tended to be higher (P = 0.07) in BBD than in normal breast, and 17β-hydroxysteroid dehydrogenase type 1 immunoreactivity was significantly higher in BBD than normal breast in the postmenopausal tissues. Immunoreactivity of estrogen and progesterone receptors was also significantly higher in BBD than normal breast. These results suggest that tissue concentration of estradiol is increased in BBD at a level similar to DCIS, which is considered mainly due to loss of estrogen sulfotransferase expression. Increased local estradiol concentration in BBD due to aberrant expression of estrogen-producing/metabolizing enzymes may play important roles in the accumulation of estradiol-mediated growth and/or subsequent development of breast carcinoma.
© 2010 Japanese Cancer Association.
Figures


Similar articles
-
17 beta-Hydroxysteroid dehydrogenase type 1 and type 2 in ductal carcinoma in situ and intraductal proliferative lesions of the human breast.Anticancer Res. 2000 Mar-Apr;20(2B):1101-8. Anticancer Res. 2000. PMID: 10810403
-
New development in intracrinology of breast carcinoma.Breast Cancer. 2006;13(2):129-36. doi: 10.2325/jbcs.13.129. Breast Cancer. 2006. PMID: 16755106 Review.
-
Intracrinology of sex steroids in ductal carcinoma in situ (DCIS) of human breast: comparison to invasive ductal carcinoma (IDC) and non-neoplastic breast.J Steroid Biochem Mol Biol. 2009 Mar;114(1-2):68-71. doi: 10.1016/j.jsbmb.2008.12.021. J Steroid Biochem Mol Biol. 2009. PMID: 19444935 Review.
-
Intratumoral concentration of sex steroids and expression of sex steroid-producing enzymes in ductal carcinoma in situ of human breast.Endocr Relat Cancer. 2008 Mar;15(1):113-24. doi: 10.1677/ERC-07-0092. Endocr Relat Cancer. 2008. PMID: 18310280
-
Recent insight on the control of enzymes involved in estrogen formation and transformation in human breast cancer.J Steroid Biochem Mol Biol. 2005 Feb;93(2-5):221-36. doi: 10.1016/j.jsbmb.2005.02.007. J Steroid Biochem Mol Biol. 2005. PMID: 15860265 Review.
Cited by
-
Peri-foci adipose-derived stem cells promote chemoresistance in breast cancer.Stem Cell Res Ther. 2017 Jul 27;8(1):177. doi: 10.1186/s13287-017-0630-2. Stem Cell Res Ther. 2017. PMID: 28750689 Free PMC article.
-
Regulation of Stromal Cells by Sex Steroid Hormones in the Breast Cancer Microenvironment.Cancers (Basel). 2024 Dec 2;16(23):4043. doi: 10.3390/cancers16234043. Cancers (Basel). 2024. PMID: 39682229 Free PMC article. Review.
-
Adipose-Derived Stem Cells in Novel Approaches to Breast Reconstruction: Their Suitability for Tissue Engineering and Oncological Safety.Breast Cancer (Auckl). 2017 Aug 16;11:1178223417726777. doi: 10.1177/1178223417726777. eCollection 2017. Breast Cancer (Auckl). 2017. PMID: 29104428 Free PMC article. Review.
-
Tongshu capsule down-regulates the expression of estrogen receptor α and suppresses human breast cancer cell proliferation.PLoS One. 2014 Aug 7;9(8):e104261. doi: 10.1371/journal.pone.0104261. eCollection 2014. PLoS One. 2014. PMID: 25101695 Free PMC article.
-
Clinical characteristics of breast cancers in African-American women with benign breast disease: a comparison to the surveillance, epidemiology, and end results program.Breast J. 2014 Nov-Dec;20(6):571-7. doi: 10.1111/tbj.12331. Epub 2014 Sep 8. Breast J. 2014. PMID: 25200244 Free PMC article.
References
-
- Devitt JE. Clinical benign disorders of the breast and carcinoma of the breast. Surg Gynecol Obstet 1981; 152: 437–40. - PubMed
-
- Hartmann LC, Sellers TA, Frost MH et al. Benign breast disease and the risk of breast cancer. N Engl J Med 2005; 353: 229–37. - PubMed
-
- Dupont WD, Page DL. Risk factors for breast cancer in women with proliferative breast disease. N Engl J Med 1985; 312: 146–51. - PubMed
-
- Carter CL, Corle DK, Micozzi MS, Schatzkin A, Taylor PR. A prospective study of the development of breast cancer in 16,692 women with benign breast disease. Am J Epidemiol 1988; 128: 467–77. - PubMed
-
- Allegra JC, Lippman ME, Green L et al. Estrogen receptor values in patients with benign breast disease. Cancer 1979; 44: 228–31. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

