Linking the acetylcholine receptor-channel agonist-binding sites with the gate
- PMID: 20682257
- PMCID: PMC2913195
- DOI: 10.1016/j.bpj.2010.05.008
Linking the acetylcholine receptor-channel agonist-binding sites with the gate
Abstract
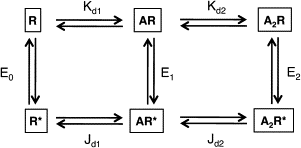
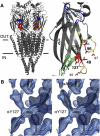
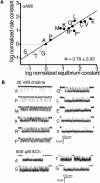
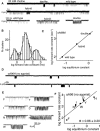
The gating isomerization of neuromuscular acetylcholine receptors links the rearrangements of atoms at two transmitter-binding sites with those at a distant gate region in the pore. To explore the mechanism of this reversible process, we estimated the gating rate and equilibrium constants for receptors with point mutations of alpha-subunit residues located between the binding sites and the membrane domain (N95, A96, Y127, and I49). The maximum energy change caused by a side-chain substitution at alphaA96 was huge (approximately 8.6 kcal/mol, the largest value measured so far for any alpha-subunit amino acid). A Phi-value analysis suggests that alphaA96 experiences its change in energy (structure) approximately synchronously with residues alphaY127 and alphaI49, but after the agonist molecule and other residues in loop A. Double mutant-cycle experiments show that the energy changes at alphaA96 are strongly coupled with those of alphaY127 and alphaI49. We identify a column of mutation-sensitive residues in the alpha-subunit that may be a pathway for energy transfer through the extracellular domain in the gating isomerization.
2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures


Similar articles
-
Acetylcholine receptor gating: movement in the alpha-subunit extracellular domain.J Gen Physiol. 2007 Dec;130(6):569-79. doi: 10.1085/jgp.200709858. J Gen Physiol. 2007. PMID: 18040059 Free PMC article.
-
Acetylcholine receptor channels activated by a single agonist molecule.Biophys J. 2010 May 19;98(9):1840-6. doi: 10.1016/j.bpj.2010.01.025. Biophys J. 2010. PMID: 20441747 Free PMC article.
-
Dynamics of the acetylcholine receptor pore at the gating transition state.Proc Natl Acad Sci U S A. 2005 Oct 18;102(42):15069-74. doi: 10.1073/pnas.0505090102. Epub 2005 Oct 10. Proc Natl Acad Sci U S A. 2005. PMID: 16217024 Free PMC article.
-
The gating isomerization of neuromuscular acetylcholine receptors.J Physiol. 2010 Feb 15;588(Pt 4):573-86. doi: 10.1113/jphysiol.2009.182774. Epub 2009 Nov 23. J Physiol. 2010. PMID: 19933754 Free PMC article. Review.
-
Acetylcholine receptors, between closed and open.Novartis Found Symp. 2002;245:223-34; discussion 234-9, 261-4. Novartis Found Symp. 2002. PMID: 12027011 Review.
Cited by
-
Signal transduction pathways in the pentameric ligand-gated ion channels.PLoS One. 2013 May 8;8(5):e64326. doi: 10.1371/journal.pone.0064326. Print 2013. PLoS One. 2013. PMID: 23667707 Free PMC article.
-
Correlating structural and energetic changes in glycine receptor activation.J Biol Chem. 2015 Feb 27;290(9):5621-34. doi: 10.1074/jbc.M114.616573. Epub 2015 Jan 8. J Biol Chem. 2015. PMID: 25572390 Free PMC article.
-
Structural link between γ-aminobutyric acid type A (GABAA) receptor agonist binding site and inner β-sheet governs channel activation and allosteric drug modulation.J Biol Chem. 2012 Feb 24;287(9):6714-24. doi: 10.1074/jbc.M111.316836. Epub 2012 Jan 4. J Biol Chem. 2012. PMID: 22219195 Free PMC article.
-
Unmasking coupling between channel gating and ion permeation in the muscle nicotinic receptor.Elife. 2021 Apr 6;10:e66225. doi: 10.7554/eLife.66225. Elife. 2021. PMID: 33821794 Free PMC article.
-
Cyclic activation of endplate acetylcholine receptors.Proc Natl Acad Sci U S A. 2017 Nov 7;114(45):11914-11919. doi: 10.1073/pnas.1711228114. Epub 2017 Oct 23. Proc Natl Acad Sci U S A. 2017. PMID: 29078356 Free PMC article.
References
-
- Changeux J.P., Devillers-Thiery A., Chemouilli P. Acetylcholine receptor: an allosteric protein. Science. 1984;225:1335–1345. - PubMed
-
- Lester H.A., Dibas M.I., Dougherty D.A. Cys-loop receptors: new twists and turns. Trends Neurosci. 2004;27:329–336. - PubMed
-
- Sine S.M., Engel A.G. Recent advances in Cys-loop receptor structure and function. Nature. 2006;440:448–455. - PubMed
-
- Monod J., Wyman J., Changeux J.P. On the nature of allosteric transitions: a plausible model. J. Mol. Biol. 1965;12:88–118. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

