INS-1 cells undergoing caspase-dependent apoptosis enhance the regenerative capacity of neighboring cells
- PMID: 20682686
- PMCID: PMC2963538
- DOI: 10.2337/db09-1478
INS-1 cells undergoing caspase-dependent apoptosis enhance the regenerative capacity of neighboring cells
Abstract
Objective: In diabetes, β-cell mass is not static but in a constant process of cell death and renewal. Inactivating mutations in transcription factor 1 (tcf-1)/hepatocyte nuclear factor1a (hnf1a) result in decreased β-cell mass and HNF1A-maturity onset diabetes of the young (HNF1A-MODY). Here, we investigated the effect of a dominant-negative HNF1A mutant (DN-HNF1A) induced apoptosis on the regenerative capacity of INS-1 cells.
Research design and methods: DN-HNF1A was expressed in INS-1 cells using a reverse tetracycline-dependent transactivator system. Gene(s)/protein(s) involved in β-cell regeneration were investigated by real-time quantitative RT-PCR, Western blotting, and immunohistochemistry. Pancreatic stone protein/regenerating protein (PSP/reg) serum levels in human subjects were detected by enzyme-linked immunosorbent assay.
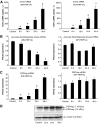
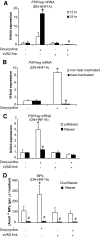
Results: We detected a prominent induction of PSP/reg at the gene and protein level during DN-HNF1A-induced apoptosis. Elevated PSP/reg levels were also detected in islets of transgenic HNF1A-MODY mice and in the serum of HNF1A-MODY patients. The induction of PSP/reg was glucose dependent and mediated by caspase activation during apoptosis. Interestingly, the supernatant from DN-HNF1A-expressing cells, but not DN-HNF1A-expressing cells treated with zVAD.fmk, was sufficient to induce PSP/reg gene expression and increase cell proliferation in naïve, untreated INS-1 cells. Further experiments demonstrated that annexin-V-positive microparticles originating from apoptosing INS-1 cells mediated the induction of PSP/reg. Treatment with recombinant PSP/reg reversed the phenotype of DN-HNF1A-induced cells by stimulating cell proliferation and increasing insulin gene expression.
Conclusions: Our results suggest that apoptosing INS-1 cells shed microparticles that may stimulate PSP/reg induction in neighboring cells, a mechanism that may facilitate the recovery of β-cell mass in HNF1A-MODY.
Figures



References
-
- Fajans SS. Scope and heterogeneous nature of MODY. Diabetes Care 1990;13:49–64 - PubMed
-
- Yamagata K, Furuta H, Oda N, Kaisaki PJ, Menzel S, Cox NJ, Fajans SS, Signorini S, Stoffel M, Bell GI. Mutations in the hepatocyte nuclear factor-4alpha gene in maturity-onset diabetes of the young (MODY1). Nature 1996;384:458–460 - PubMed
-
- Shih DQ, Screenan S, Munoz KN, Philipson L, Pontoglio M, Yaniv M, Polonsky KS, Stoffel M. Loss of HNF-1α function in mice leads to abnormal expression of genes involved in pancreatic islet development and metabolism. Diabetes 2001;50:2472–2480 - PubMed
-
- Ben-Shushan E, Marshak S, Shoshkes M, Cerasi E, Melloul D. A pancreatic beta-cell-specific enhancer in the human PDX-1 gene is regulated by hepatocyte nuclear factor 3beta (HNF-3beta), HNF-1alpha, and SPs transcription factors. J Biol Chem 2001;276:17533–17540 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

