Metastasis-associated protein 1 and its short form variant stimulates Wnt1 transcription through promoting its derepression from Six3 corepressor
- PMID: 20682799
- PMCID: PMC3711655
- DOI: 10.1158/0008-5472.CAN-10-0909
Metastasis-associated protein 1 and its short form variant stimulates Wnt1 transcription through promoting its derepression from Six3 corepressor
Abstract
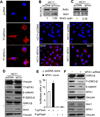
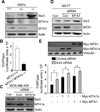
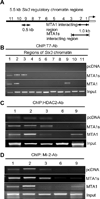
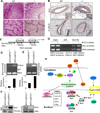
Although Wnt1 downstream signaling components have been well studied and activated in human cancer, the pathways that regulate Wnt1 itself have not been explored in depth. Here, we provide gain-of-function, loss-of function, and molecular evidence supporting functional interactions between metastasis-associated protein 1 short-form (MTA1s), metastasis-associated protein 1 (MTA1), and Wnt1 signaling components during mammary gland development and tumorigenesis. Using multiple model systems involving overexpression or knockdown of MTA1s or MTA1, we discovered that MTA1s and MTA1 hyperactivate the Wnt1 pathway due to increased expression of Wnt1 transcription. MTA1s and MTA1 physically interact with Six3 chromatin, a protein product of which is a direct histone deacetylase inhibitor-dependent repressor of Wnt1 transcription. Deletion of the MTA1s and MTA1 allele in murine embryonic fibroblasts resulted in the upregulation of Six3 and downregulation of Wnt signaling. In addition, mammary glands from the MTA1s/MTA1(-/-) mice exhibited increased recruitment of Six3 corepressor complex to the Wnt1 promoter and inhibition of Wnt1 pathway in mammary glands. These findings identify MTA1s and MTA1 as important upstream modifiers of the Wnt1 transcription, and consequently its functions, by directly inhibiting the transcription of Six3, allowing derepression of Wnt1 transcription.
(c)2010 AACR.
Figures



Similar articles
-
Leptin-induced epithelial-mesenchymal transition in breast cancer cells requires β-catenin activation via Akt/GSK3- and MTA1/Wnt1 protein-dependent pathways.J Biol Chem. 2012 Mar 9;287(11):8598-612. doi: 10.1074/jbc.M111.322800. Epub 2012 Jan 23. J Biol Chem. 2012. PMID: 22270359 Free PMC article.
-
Metastasis-associated protein 1 short form stimulates Wnt1 pathway in mammary epithelial and cancer cells.Cancer Res. 2010 Aug 15;70(16):6598-608. doi: 10.1158/0008-5472.CAN-10-0907. Cancer Res. 2010. PMID: 20710043 Free PMC article.
-
Repression of Six3 by a corepressor regulates rhodopsin expression.Proc Natl Acad Sci U S A. 2007 Aug 7;104(32):13128-33. doi: 10.1073/pnas.0705878104. Epub 2007 Jul 31. Proc Natl Acad Sci U S A. 2007. PMID: 17666527 Free PMC article.
-
Emerging roles of MTA family members in human cancers.Semin Oncol. 2003 Oct;30(5 Suppl 16):30-7. doi: 10.1053/j.seminoncol.2003.08.005. Semin Oncol. 2003. PMID: 14613024 Review.
-
The role of the MTA family and their encoded proteins in human cancers: molecular functions and clinical implications.Clin Exp Metastasis. 2009;26(3):215-27. doi: 10.1007/s10585-008-9233-8. Epub 2008 Dec 31. Clin Exp Metastasis. 2009. PMID: 19116762 Review.
Cited by
-
Leptin-induced epithelial-mesenchymal transition in breast cancer cells requires β-catenin activation via Akt/GSK3- and MTA1/Wnt1 protein-dependent pathways.J Biol Chem. 2012 Mar 9;287(11):8598-612. doi: 10.1074/jbc.M111.322800. Epub 2012 Jan 23. J Biol Chem. 2012. PMID: 22270359 Free PMC article.
-
Physiological functions of MTA family of proteins.Cancer Metastasis Rev. 2014 Dec;33(4):869-77. doi: 10.1007/s10555-014-9514-4. Cancer Metastasis Rev. 2014. PMID: 25344801 Free PMC article. Review.
-
Expression of MTA1 in nasopharyngeal carcinoma and its correlation with prognosis.Med Oncol. 2014 Dec;31(12):330. doi: 10.1007/s12032-014-0330-z. Epub 2014 Nov 22. Med Oncol. 2014. PMID: 25416046
-
MTA1--a stress response protein: a master regulator of gene expression and cancer cell behavior.Cancer Metastasis Rev. 2014 Dec;33(4):1001-9. doi: 10.1007/s10555-014-9525-1. Cancer Metastasis Rev. 2014. PMID: 25332145 Free PMC article. Review.
-
The plant alkaloid tetrandrine inhibits metastasis via autophagy-dependent Wnt/β-catenin and metastatic tumor antigen 1 signaling in human liver cancer cells.J Exp Clin Cancer Res. 2018 Jan 15;37(1):7. doi: 10.1186/s13046-018-0678-6. J Exp Clin Cancer Res. 2018. PMID: 29334999 Free PMC article.
References
-
- Tsukamoto AS, Grosschedl R, Guzman RC, Parslow T, Varmus HE. Expression of the int-1 gene in transgenic mice is associated with mammary gland hyperplasia and adenocarcinomas in male and female mice. Cell. 1988;55(4):619–625. - PubMed
-
- Moon RT, Bowerman B, Boutros M, Perrimon N. The promise and perils of wnt signaling through beta-catenin. Science. 2002;296(5573):1644–1646. - PubMed
-
- Zhu CC, Dyer MA, Uchikawa M, Kondoh H, Lagutin OV, Oliver G. Six3-mediated auto repression and eye development requires its interaction with members of the groucho-related family of co-repressors. Development. 2002;129(12):2835–2849. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

