The vascular contribution to Alzheimer's disease
- PMID: 20684749
- PMCID: PMC2950620
- DOI: 10.1042/CS20100094
The vascular contribution to Alzheimer's disease
Abstract
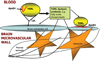
AD (Alzheimer's disease) is a progressive neurodegenerative disease of unknown origin. Despite questions as to the underlying cause(s) of this disease, shared risk factors for both AD and atherosclerotic cardiovascular disease indicate that vascular mechanisms may critically contribute to the development and progression of both AD and atherosclerosis. An increased risk of developing AD is linked to the presence of the apoE4 (apolipoprotein E4) allele, which is also strongly associated with increased risk of developing atherosclerotic cardiovascular disease. Recent studies also indicate that cardiovascular risk factors, including elevated blood cholesterol and triacylglycerol (triglyceride), increase the likelihood of AD and vascular dementia. Lipids and lipoproteins in the circulation interact intimately with the cerebrovasculature, and may have important effects on its constituent brain microvascular endothelial cells and the adjoining astrocytes, which are components of the neurovascular unit. The present review will examine the potential mechanisms for understanding the contributions of vascular factors, including lipids, lipoproteins and cerebrovascular Abeta (amyloid beta), to AD, and suggest therapeutic strategies for the attenuation of this devastating disease process. Specifically, we will focus on the actions of apoE, TGRLs (triacylglycerol-rich lipoproteins) and TGRL lipolysis products on injury of the neurovascular unit and increases in blood-brain barrier permeability.
Figures


References
-
- Aggarwal NT, Decarli C. Vascular dementia: emerging trends. Semin. Neurol. 2007;27:66–77. - PubMed
-
- DeCarli C. The role of cerebrovascular disease in dementia. Neurologist. 2003;9:123–136. - PubMed
-
- Berglund L, Wiklund O, Eggertsen G, Olofsson SO, Eriksson M, Linden T, Bondjers G, Angelin B. Apolipoprotein E phenotypes in familial hypercholesterolaemia: importance for expression of disease and response to therapy. J. Intern. Med. 1993;233:173–178. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

