Cardiac muscle ring finger-1--friend or foe?
- PMID: 20685572
- PMCID: PMC2917387
- DOI: 10.1016/j.tcm.2010.03.001
Cardiac muscle ring finger-1--friend or foe?
Abstract
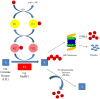
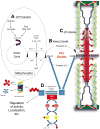
The ubiquitin proteasome system plays a role in regulating protein activity and is integral to the turnover of damaged and worn proteins. In this review, we discuss the recently described relationship between the ubiquitin proteasome system and the cardiac creatine kinase/phosphocreatine shuttle, an essential component of adenosine triphosphate generation and energy shuttling within the heart. The ubiquitin ligase muscle ring finger-1 (MuRF1) binds creatine kinase, leading to its ubiquitination and possible degradation. Muscle ring finger-1 may also be integral in the regulation of creatine kinase activity in vivo. Because there is a close relationship between the cardiac creatine kinase/phosphocreatine shuttle activity and heart failure, these findings suggest that MuRF1's role in protein quality control of creatine kinase may be vital to the regulation and maintenance of cardiac energetics to protect against heart failure.
(c) 2010 Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures
Similar articles
-
The ubiquitin ligase MuRF1 protects against cardiac ischemia/reperfusion injury by its proteasome-dependent degradation of phospho-c-Jun.Am J Pathol. 2011 Mar;178(3):1043-58. doi: 10.1016/j.ajpath.2010.11.049. Am J Pathol. 2011. PMID: 21356357 Free PMC article.
-
Muscle RING-finger protein-1 (MuRF1) as a connector of muscle energy metabolism and protein synthesis.J Mol Biol. 2008 Mar 7;376(5):1224-36. doi: 10.1016/j.jmb.2007.11.049. Epub 2007 Nov 22. J Mol Biol. 2008. PMID: 18222470
-
Cardiac proteasome activity in muscle ring finger-1 null mice at rest and following synthetic glucocorticoid treatment.Am J Physiol Endocrinol Metab. 2011 Nov;301(5):E967-77. doi: 10.1152/ajpendo.00165.2011. Epub 2011 Aug 9. Am J Physiol Endocrinol Metab. 2011. PMID: 21828340 Free PMC article.
-
Regulation of AMPK by the ubiquitin proteasome system.Am J Pathol. 2011 Jan;178(1):4-11. doi: 10.1016/j.ajpath.2010.11.030. Epub 2010 Dec 23. Am J Pathol. 2011. PMID: 21224036 Free PMC article. Review.
-
A systematic view on E3 ligase Ring TRIMmers with a focus on cardiac function and disease.Trends Cardiovasc Med. 2019 Jan;29(1):1-8. doi: 10.1016/j.tcm.2018.05.007. Epub 2018 May 28. Trends Cardiovasc Med. 2019. PMID: 29880235
Cited by
-
Cardiac systolic dysfunction in doxorubicin-challenged rats is associated with upregulation of MuRF2 and MuRF3 E3 ligases.Exp Clin Cardiol. 2012 Sep;17(3):101-9. Exp Clin Cardiol. 2012. PMID: 23620696 Free PMC article.
-
Cytoskeletal protein kinases: titin and its relations in mechanosensing.Pflugers Arch. 2011 Jul;462(1):119-34. doi: 10.1007/s00424-011-0946-1. Epub 2011 Mar 18. Pflugers Arch. 2011. PMID: 21416260 Free PMC article. Review.
-
Experimental ischaemic stroke induces transient cardiac atrophy and dysfunction.J Cachexia Sarcopenia Muscle. 2019 Feb;10(1):54-62. doi: 10.1002/jcsm.12335. Epub 2018 Oct 30. J Cachexia Sarcopenia Muscle. 2019. PMID: 30378296 Free PMC article.
-
Generation of MuRF-GFP transgenic zebrafish models for investigating murf gene expression and protein localization in Smyd1b and Hsp90α1 knockdown embryos.Comp Biochem Physiol B Biochem Mol Biol. 2020 Feb;240:110368. doi: 10.1016/j.cbpb.2019.110368. Epub 2019 Oct 24. Comp Biochem Physiol B Biochem Mol Biol. 2020. PMID: 31669374 Free PMC article.
-
Developmental regulation of MURF E3 ubiquitin ligases in skeletal muscle.J Muscle Res Cell Motil. 2012 Jun;33(2):107-22. doi: 10.1007/s10974-012-9288-7. Epub 2012 Mar 17. J Muscle Res Cell Motil. 2012. PMID: 22426552 Free PMC article.
References
-
- Adams V, Linke A, Gielen S, et al. Modulation of Murf-1 and MAFbx expression in the myocardium by physical exercise training. Eur J Cardiovasc Prev Rehabil. 2008;15:293–299. - PubMed
-
- Boehm E, Ventura-Clapier R, Mateo P, et al. Glycolysis supports calcium uptake by the sarcoplasmic reticulum in skinned ventricular fibres of mice deficient in mitochondrial and cytosolic creatine kinase. J Mol Cell Cardiol. 2000;32:891–902. - PubMed
-
- Dieni CA, Storey KB. Creatine kinase regulation by reversible phosphorylation in frog muscle. Comp Biochem Physiol B Biochem Mol Biol. 2009;152:405–412. - PubMed
-
- Dunlop RA, Brunk UT, Rodgers KJ. Oxidized proteins: Mechanisms of removal and consequences of accumulation. IUBMB Life. 2009;61:522–527. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

