Long and short multifunicular projections of sacral neurons are activated by sensory input to produce locomotor activity in the absence of supraspinal control
- PMID: 20685976
- PMCID: PMC6634674
- DOI: 10.1523/JNEUROSCI.1208-10.2010
Long and short multifunicular projections of sacral neurons are activated by sensory input to produce locomotor activity in the absence of supraspinal control
Abstract
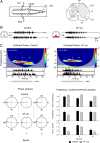
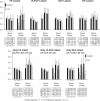
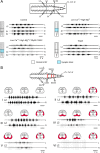
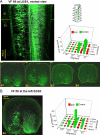
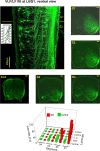
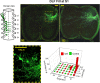
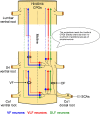
Afferent input from load and joint receptors has been shown to reactivate the central pattern generators for locomotion (CPGs) in spinal cord injury patients and thereby improve their motor function and mobility. Elucidation of the pathways interposed between the afferents and CPGs is critical for the determination of the capacity of sensory input to activate the CPGs when the continuity of the white matter tracts is impaired following spinal cord injury. Using electrophysiological recordings, confocal imaging studies of spinal neurons and surgical manipulations of the white matter, we show that the capacity of sacrocaudal afferent (SCA) input to produce locomotor activity in isolated rat spinal cords depends not only on long ascending pathways, but also on recruitment of sacral proprioneurons interposed between the second order neurons and the hindlimb CPGs. We argue that large heterogeneous populations of second-order and proprioneurons whose crossed and uncrossed axons project rostrally through the ventral, ventrolateral/lateral and dorsolateral white matter funiculi contribute to the generation of the rhythm by the stimulated sacrocaudal input. The complex organization and multiple projection patterns of these populations enable the sacrocaudal afferent input to activate the CPGs even if the white matter pathways are severely damaged. Further studies are required to clarify the mechanisms involved in SCA-induced locomotor activity and assess its potential use for the rescue of lost motor functions after spinal cord injury.
Figures


Similar articles
-
Characterization of sacral interneurons that mediate activation of locomotor pattern generators by sacrocaudal afferent input.J Neurosci. 2013 Jan 9;33(2):734-47. doi: 10.1523/JNEUROSCI.4390-12.2013. J Neurosci. 2013. PMID: 23303951 Free PMC article.
-
Sensory-induced activation of pattern generators in the absence of supraspinal control.Ann N Y Acad Sci. 2010 Jun;1198:54-62. doi: 10.1111/j.1749-6632.2009.05424.x. Ann N Y Acad Sci. 2010. PMID: 20536920
-
Shaping the Output of Lumbar Flexor Motoneurons by Sacral Neuronal Networks.J Neurosci. 2017 Feb 1;37(5):1294-1311. doi: 10.1523/JNEUROSCI.2213-16.2016. Epub 2016 Dec 26. J Neurosci. 2017. PMID: 28025254 Free PMC article.
-
The sacral networks and neural pathways used to elicit lumbar motor rhythm in the rodent spinal cord.Front Neural Circuits. 2014 Dec 3;8:143. doi: 10.3389/fncir.2014.00143. eCollection 2014. Front Neural Circuits. 2014. PMID: 25520624 Free PMC article. Review.
-
Ascending pathways that mediate cholinergic modulation of lumbar motor activity.J Neurochem. 2017 Aug;142 Suppl 2:82-89. doi: 10.1111/jnc.14065. J Neurochem. 2017. PMID: 28791705 Review.
Cited by
-
Inducing hindlimb locomotor recovery in adult rat after complete thoracic spinal cord section using repeated treadmill training with perineal stimulation only.J Neurophysiol. 2015 Sep;114(3):1931-46. doi: 10.1152/jn.00416.2015. Epub 2015 Jul 22. J Neurophysiol. 2015. PMID: 26203108 Free PMC article.
-
Improvement in Motor and Walking Capacity during Multisegmental Transcutaneous Spinal Stimulation in Individuals with Incomplete Spinal Cord Injury.Int J Mol Sci. 2024 Apr 19;25(8):4480. doi: 10.3390/ijms25084480. Int J Mol Sci. 2024. PMID: 38674065 Free PMC article.
-
Electrical epidural stimulation of the cervical spinal cord: implications for spinal respiratory neuroplasticity after spinal cord injury.J Neurophysiol. 2021 Aug 1;126(2):607-626. doi: 10.1152/jn.00625.2020. Epub 2021 Jul 7. J Neurophysiol. 2021. PMID: 34232771 Free PMC article. Review.
-
5-HT₂ and 5-HT₇ receptor agonists facilitate plantar stepping in chronic spinal rats through actions on different populations of spinal neurons.Front Neural Circuits. 2014 Aug 19;8:95. doi: 10.3389/fncir.2014.00095. eCollection 2014. Front Neural Circuits. 2014. PMID: 25191231 Free PMC article.
-
Unique Spatiotemporal Neuromodulation of the Lumbosacral Circuitry Shapes Locomotor Success after Spinal Cord Injury.J Neurotrauma. 2016 Sep 15;33(18):1709-23. doi: 10.1089/neu.2015.4256. Epub 2016 Apr 20. J Neurotrauma. 2016. PMID: 26792233 Free PMC article.
References
-
- Arsénio Nunes ML, Sotelo C. Development of the spinocerebellar system in the postnatal rat. J Comp Neurol. 1985;237:291–306. - PubMed
-
- Baldissera F, Hultborn H, Illert M. Integration in spinal neuronal systems. In: Brooks VB, editor. Handbook of Physiology. Bethesda, MD: American Physiological Society; 1981. pp. 509–595.
-
- Blivis D, Mentis GZ, O'Donovan MJ, Lev-Tov A. Differential effects of opioids on sacrocaudal afferent pathways and central pattern generators in the neonatal rat spinal cord. J Neurophysiol. 2007;97:2875–2886. - PubMed
-
- Blivis D, Mentis GZ, O'Donovan MJ, Lev-Tov A. Studies of sacral neurons involved in activation of the lumbar central pattern generator for locomotion in the neonatal rodent spinal cord. Soc Neurosci Abstr. 2009;35:564–8.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
