Racemisation and human cataract. D-Ser, D-Asp/Asn and D-Thr are higher in the lifelong proteins of cataract lenses than in age-matched normal lenses
- PMID: 20686926
- PMCID: PMC3127471
- DOI: 10.1007/s11357-010-9171-7
Racemisation and human cataract. D-Ser, D-Asp/Asn and D-Thr are higher in the lifelong proteins of cataract lenses than in age-matched normal lenses
Abstract
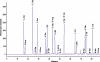
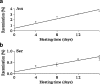
ASTRACT: Several amino acids were found to undergo progressive age-dependent racemisation in the lifelong proteins of normal human lenses. The two most highly racemised were Ser and Asx. By age 70, 4.5% of all Ser residues had been racemised, along with >9% of Asx residues. Such a high level of inversion, equivalent to between 2 and 3 D- amino acids per polypeptide chain, is likely to induce significant denaturation of the crystallins in aged lenses. Thr, Glx and Phe underwent age-dependent racemisation to a smaller degree. In model experiments, D- amino acid content could be increased simply by exposing intact lenses to elevated temperature. In cataract lenses, the extent of racemisation of Ser, Asx and Thr residues was significantly greater than for age-matched normal lenses. This was true, even for cataract lenses removed from patients at the earliest ages where age-related cataract is observed clinically. Racemisation of amino acids in crystallins may arise due to prolonged exposure of these proteins to ocular temperatures and increased levels of racemisation may play a significant role in the opacification of human lenses.
Figures



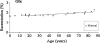
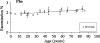

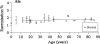
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
