Total synthesis of fostriecin: via a regio- and stereoselective polyene hydration, oxidation, and hydroboration sequence
- PMID: 20687585
- PMCID: PMC2929272
- DOI: 10.1021/ol101340n
Total synthesis of fostriecin: via a regio- and stereoselective polyene hydration, oxidation, and hydroboration sequence
Abstract
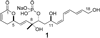
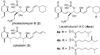
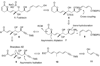
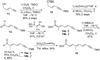
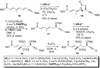
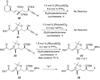
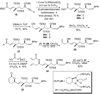
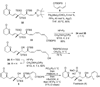
A total synthesis of the fostriecin has been achieved in 24 steps from enyne 11. The lactone moiety was installed by a Leighton allylation and Grubbs ring-closing metathesis reaction. The highly reactive Z,Z,E-triene moiety was installed via a late-stage Suzuki-Miyaura cross-coupling of a remarkably stable Z-vinyl boronate. The relative and absolute stereocenters of the C-8,9,11 triol were generated with a regio- and stereoselective asymmetric hydration/oxidation sequence.
Figures
Similar articles
-
Synthesis of Dehydro-Dephospho-Fostriecin and Formal Total Synthesis of Fostriecin.Org Lett. 2019 Oct 18;21(20):8334-8338. doi: 10.1021/acs.orglett.9b03120. Epub 2019 Oct 4. Org Lett. 2019. PMID: 31584287
-
Stereoselective Total Synthesis of Atractylodemayne A, a Conjugated 2(E),8(Z),10(E)-Triene-4,6-diyne.Org Lett. 2016 Mar 4;18(5):1162-5. doi: 10.1021/acs.orglett.6b00274. Epub 2016 Feb 17. Org Lett. 2016. PMID: 26886865
-
Formal total synthesis of fostriecin by 1,4-asymmetric induction with an alkyne-cobalt complex.Chemistry. 2010 Sep 3;16(33):10150-9. doi: 10.1002/chem.201000795. Chemistry. 2010. PMID: 20645347
-
Synthetic Strategies Employed for the Construction of Fostriecin and Related Natural Products.Chem Rev. 2016 Dec 28;116(24):15035-15088. doi: 10.1021/acs.chemrev.6b00488. Epub 2016 Dec 8. Chem Rev. 2016. PMID: 28027648 Free PMC article. Review.
-
De novo synthesis of natural products via the asymmetric hydration of polyenes.Chem Commun (Camb). 2011 Aug 14;47(30):8493-505. doi: 10.1039/c1cc11791b. Epub 2011 May 11. Chem Commun (Camb). 2011. PMID: 21559534 Free PMC article. Review.
Cited by
-
Synthesis of Z-(pinacolato)allylboron and Z-(pinacolato)alkenylboron compounds through stereoselective catalytic cross-metathesis.J Am Chem Soc. 2013 Apr 24;135(16):6026-9. doi: 10.1021/ja403188t. Epub 2013 Apr 15. J Am Chem Soc. 2013. PMID: 23586708 Free PMC article.
-
Synthesis of cyclic alkenylsiloxanes by semihydrogenation: a stereospecific route to (Z)-alkenyl polyenes.Chemistry. 2014 Jul 7;20(28):8594-8. doi: 10.1002/chem.201403255. Epub 2014 Jun 4. Chemistry. 2014. PMID: 24899240 Free PMC article.
-
A Highly Convergent Total Synthesis of Leustroducsin B.J Am Chem Soc. 2015 Sep 16;137(36):11594-7. doi: 10.1021/jacs.5b07438. Epub 2015 Sep 1. J Am Chem Soc. 2015. PMID: 26313159 Free PMC article.
-
[Pt(PPh3)4]-Catalyzed Selective Diboration of Symmetrical and Unsymmetrical 1,3-Diynes.J Org Chem. 2022 Aug 19;87(16):10651-10663. doi: 10.1021/acs.joc.2c00844. Epub 2022 Aug 2. J Org Chem. 2022. PMID: 35917577 Free PMC article.
-
Formal Synthesis of Fostriecin via Asymmetric Alcohol-Mediated Carbonyl Allylation.Org Lett. 2025 May 2;27(17):4501-4506. doi: 10.1021/acs.orglett.5c01026. Epub 2025 Apr 10. Org Lett. 2025. PMID: 40209063 Free PMC article.
References
-
-
Reviews: Jackson RC, Fry DW, Boritzki TJ, Roberts BJ, Hook KE, Leopold WR. Adv. Enzyme Regul. 1985;23:193. Scheithauer W, Hoff DDV, Clark GM, Shillis JL, Elslager EF. Eur. J. Clin. Oncol. 1986;22:921.
-
-
-
Reviews: Walsh AH, Cheng A, Honkanen RE. FEBS Lett. 1997;416:230. Hastie CJ, Cohen PTW. FEBS Lett. 1998;431:357.
-
-
- Boger DL, Hikota M, Lewis BM. J. Org. Chem. 1997;62:1748.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

