Physiological effects and mechanisms of action of endocrine disrupting chemicals that alter estrogen signaling
- PMID: 20688617
- PMCID: PMC4782146
- DOI: 10.14310/horm.2002.1270
Physiological effects and mechanisms of action of endocrine disrupting chemicals that alter estrogen signaling
Figures
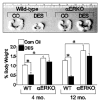
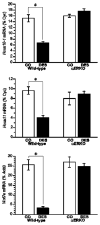
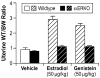
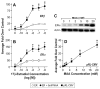
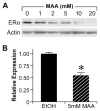

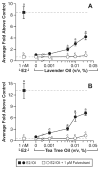
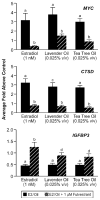
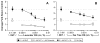
Similar articles
-
Endocrine-disrupting chemicals use distinct mechanisms of action to modulate endocrine system function.Endocrinology. 2006 Jun;147(6 Suppl):S25-32. doi: 10.1210/en.2005-1117. Epub 2006 May 11. Endocrinology. 2006. PMID: 16690802 Review.
-
Effects of Diethylstilbestrol on Zebrafish Gonad Development and Endocrine Disruption Mechanism.Biomolecules. 2021 Jun 25;11(7):941. doi: 10.3390/biom11070941. Biomolecules. 2021. PMID: 34201983 Free PMC article.
-
Chemicals with endocrine-disrupting potential: a threat to human health?Angew Chem Int Ed Engl. 2005 Sep 5;44(35):5568-74. doi: 10.1002/anie.200502138. Angew Chem Int Ed Engl. 2005. PMID: 16136609 No abstract available.
-
[Xenoestrogens: endocrine disrupting compounds].Ginekol Pol. 2008 Nov;79(11):785-90. Ginekol Pol. 2008. PMID: 19140503 Review. Polish.
-
Endocrine disrupting effects of zearalenone, alpha- and beta-zearalenol at the level of nuclear receptor binding and steroidogenesis.Toxicol Lett. 2011 Oct 10;206(2):210-7. doi: 10.1016/j.toxlet.2011.07.015. Epub 2011 Jul 23. Toxicol Lett. 2011. PMID: 21803136
Cited by
-
Differential in Vitro Biological Action, Coregulator Interactions, and Molecular Dynamic Analysis of Bisphenol A (BPA), BPAF, and BPS Ligand-ERα Complexes.Environ Health Perspect. 2018 Jan 31;126(1):017012. doi: 10.1289/EHP2505. Environ Health Perspect. 2018. PMID: 29389661 Free PMC article.
-
Drugs and Medical Devices: Adverse Events and the Impact on Women's Health.Clin Ther. 2017 Jan;39(1):10-22. doi: 10.1016/j.clinthera.2016.12.009. Epub 2017 Jan 7. Clin Ther. 2017. PMID: 28069260 Free PMC article. Review.
-
Fingerprinting of neurotoxic compounds using a mouse embryonic stem cell dual luminescence reporter assay.Arch Toxicol. 2017 Jan;91(1):365-391. doi: 10.1007/s00204-016-1690-2. Epub 2016 Mar 25. Arch Toxicol. 2017. PMID: 27015953 Free PMC article.
-
Activation of Estrogen Receptor by Bavachin from Psoralea corylifolia.Biomol Ther (Seoul). 2012 Mar;20(2):183-8. doi: 10.4062/biomolther.2012.20.2.183. Biomol Ther (Seoul). 2012. PMID: 24116293 Free PMC article.
-
Estrogen receptor is activated by korean red ginseng in vitro but not in vivo.J Ginseng Res. 2012 Apr;36(2):169-75. doi: 10.5142/jgr.2012.36.2.169. J Ginseng Res. 2012. PMID: 23717117 Free PMC article.
References
-
- Dodds EC, Lawsaon W, Robinson R. Estrogenic activity of certain synthetic compounds. Nature. 1938:141247–141248.
-
- Smith OW, Smith GBS, Hurwitz D. Increased excretion of pregnanediol in pregnancy from diethylstilbestrol with special reference to the prevention of late pregnancy accidents. Med Rec Ann. 1946;40:1669–1671. - PubMed
-
- Dieckmann WJ, Davis ME, Rynkiewicz LM, Pottinger RE, Gabbe SG. Does the administration of diethylstilbestrol during pregnancy have therapeutic value 1953? Am J Obstet Gynecol. 1999;66:1062–1081. - PubMed
-
- Herbst AL, Ulfelder H, Poskanzer DC. Adenocarcinoma of the vagina. Association of maternal stilbestrol therapy with tumor appearance in young women. N Engl J Med. 1971;284:878–881. - PubMed
-
- Melnick S, Cole P, Anderson D, Herbst A. Rates and risks of diethylstilbestrol-related clear-cell adenocarcinoma of the vagina and cervix. An update. N Engl J Med. 1987;316:514–516. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

