HIF-1 mediates pathogenic inflammatory responses to intestinal ischemia-reperfusion injury
- PMID: 20689059
- PMCID: PMC2957330
- DOI: 10.1152/ajpgi.00065.2010
HIF-1 mediates pathogenic inflammatory responses to intestinal ischemia-reperfusion injury
Abstract
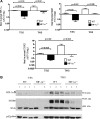
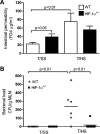
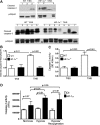
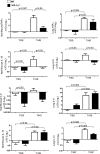
Acute lung injury (ALI) and the development of the multiple organ dysfunction syndrome (MODS) are major causes of death in trauma patients. Gut inflammation and loss of gut barrier function as a consequence of splanchnic ischemia-reperfusion (I/R) have been implicated as the initial triggering events that contribute to the development of the systemic inflammatory response, ALI, and MODS. Since hypoxia-inducible factor (HIF-1) is a key regulator of the physiological and pathophysiological response to hypoxia, we asked whether HIF-1 plays a proximal role in the induction of gut injury and subsequent lung injury. Utilizing partially HIF-1α-deficient mice in a global trauma hemorrhagic shock (T/HS) model, we found that HIF-1 activation was necessary for the development of gut injury and that the prevention of gut injury was associated with an abrogation of lung injury. Specifically, in vivo studies demonstrated that partial HIF-1α deficiency ameliorated T/HS-induced increases in intestinal permeability, bacterial translocation, and caspase-3 activation. Lastly, partial HIF-1α deficiency reduced TNF-α, IL-1β, cyclooxygenase-2, and inducible nitric oxide synthase levels in the ileal mucosa after T/HS whereas IL-1β mRNA levels were reduced in the lung after T/HS. This study indicates that prolonged intestinal HIF-1 activation is a proximal regulator of I/R-induced gut mucosal injury and gut-induced lung injury. Consequently, these results provide unique information on the initiating events in trauma-hemorrhagic shock-induced ALI and MODS as well as potential therapeutic insights.
Figures



Similar articles
-
Hypoxia-inducible factor plays a gut-injurious role in intestinal ischemia reperfusion injury.Am J Physiol Gastrointest Liver Physiol. 2011 May;300(5):G853-61. doi: 10.1152/ajpgi.00459.2010. Epub 2010 Dec 23. Am J Physiol Gastrointest Liver Physiol. 2011. PMID: 21183660 Free PMC article.
-
Inhibition of hypoxia inducible factor-1α ameliorates lung injury induced by trauma and hemorrhagic shock in rats.Acta Pharmacol Sin. 2012 May;33(5):635-43. doi: 10.1038/aps.2012.5. Epub 2012 Apr 2. Acta Pharmacol Sin. 2012. PMID: 22465950 Free PMC article.
-
Critical role of toll-like receptor 4 in hypoxia-inducible factor 1α activation during trauma/hemorrhagic shocky induced acute lung injury after lymph infusion in mice.Shock. 2014 Sep;42(3):271-8. doi: 10.1097/SHK.0000000000000212. Shock. 2014. PMID: 24978883
-
[Induction mechanism of shock: applying the etiology in judgment of the cause of death in forensic practice].Nihon Hoigaku Zasshi. 2004 Sep;58(2):130-40. Nihon Hoigaku Zasshi. 2004. PMID: 15526767 Review. Japanese.
-
Hypoxia signaling during intestinal ischemia and inflammation.Curr Opin Crit Care. 2012 Apr;18(2):178-85. doi: 10.1097/MCC.0b013e3283514bd0. Curr Opin Crit Care. 2012. PMID: 22322265 Free PMC article. Review.
Cited by
-
Intestinal epithelial cell-derived μ-opioid signaling protects against ischemia reperfusion injury through PI3K signaling.Am J Pathol. 2013 Mar;182(3):776-85. doi: 10.1016/j.ajpath.2012.11.021. Epub 2013 Jan 2. Am J Pathol. 2013. PMID: 23291213 Free PMC article.
-
The HIF-1/glial TIM-3 axis controls inflammation-associated brain damage under hypoxia.Nat Commun. 2015 Mar 20;6:6340. doi: 10.1038/ncomms7340. Nat Commun. 2015. PMID: 25790768 Free PMC article.
-
A Novel Role of OS-9 in the Maintenance of Intestinal Barrier Function from Hypoxia-induced Injury via p38-dependent Pathway.Int J Biol Sci. 2015 Apr 27;11(6):664-71. doi: 10.7150/ijbs.10783. eCollection 2015. Int J Biol Sci. 2015. PMID: 25999789 Free PMC article.
-
Hypoxia signaling in the equine small intestine: Expression and distribution of hypoxia inducible factors during experimental ischemia.Front Vet Sci. 2023 Feb 24;10:1110019. doi: 10.3389/fvets.2023.1110019. eCollection 2023. Front Vet Sci. 2023. PMID: 36908508 Free PMC article.
-
(-)-Epigallocatechin-3-gallate promotes intestinal epithelial proliferation and barrier function after ischemia/reperfusion injury via activation of Nurr1.Pharm Biol. 2023 Dec;61(1):1310-1317. doi: 10.1080/13880209.2023.2245445. Pharm Biol. 2023. PMID: 37621064 Free PMC article.
References
-
- Bakker WJ, Harris IS, Mak TW. FOXO3a is activated in response to hypoxic stress and inhibits HIF1-induced apoptosis via regulation of CITED2. Mol Cell 28: 941–953, 2007. - PubMed
-
- Bergeron M, Gidday JM, Yu AY, Semenza GL, Ferriero DM, Sharp FR. Role of hypoxia-inducible factor-1 in hypoxia-induced ischemic tolerance in neonatal rat brain. Ann Neurol 48: 285–296, 2000. - PubMed
-
- Bernaudin M, Nedelec A, Divoux D, MacKenzie E, Petit E, Schumann-Bard P. Normobaric hypoxia induces tolerance to focal permanent cerebral ischemia in association with an increased expression of hypoxia-inducible factor-1 and its target genes, erythropoietin and VEGF, in the adult mouse brain. J Cereb Blood Flow Metab 22: 393–403, 2002. - PubMed
-
- Bosch-Marce M, Okuyama H, Wesley J, Sarkar K, Kimura H, Liu Y, Zhang H, Strazza M, Rey S, Savino L, Zhou Y, McDonald K, Na Y, Vandiver S, Rabi A, Shaked Y, Kerbel R, LaVallee T, Semenza G. Effects of aging and hypoxia-inducible factor-1 activity on angiogenic cell mobilization and recovery of perfusion after limb ischemia. Circ Res 101: 1310–1318, 2007. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

