Eicosanoids in the innate immune response: TLR and non-TLR routes
- PMID: 20689730
- PMCID: PMC2905620
- DOI: 10.1155/2010/201929
Eicosanoids in the innate immune response: TLR and non-TLR routes
Abstract
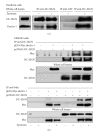
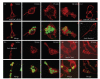
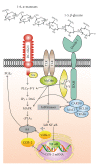
The variable array of pattern receptor expression in different cells of the innate immune system explains the induction of distinct patterns of arachidonic acid (AA) metabolism. Peptidoglycan and mannan were strong stimuli in neutrophils, whereas the fungal extract zymosan was the most potent stimulus in monocyte-derived dendritic cells since it induced the production of PGE(2), PGD(2), and several cytokines including a robust IL-10 response. Zymosan activated kappaB-binding activity, but inhibition of NF-kappaB was associated with enhanced IL-10 production. In contrast, treatments acting on CREB (CRE binding protein), including PGE(2), showed a direct correlation between CREB activation and IL-10 production. Therefore, in dendritic cells zymosan induces il10 transcription by a CRE-dependent mechanism that involves autocrine secretion of PGE(2), thus unraveling a functional cooperation between eicosanoid production and cytokine production.
Figures



References
-
- Garcia C, Montero M, Alvarez J, Sánchez Crespo M. Biosynthesis of platelet-activating factor (PAF) induced by chemotactic peptide is modulated at the lyso-PAF:acetyl-CoA acetyltransferase level by calcium transient and phosphatidic acid. Journal of Biological Chemistry. 1993;268(6):4001–4008. - PubMed
-
- Garcia Rodriguez C, Montero M, Alvarez J, García-Sancho J, Sánchez Crespo M. Dissociation of platelet-activating factor production and arachidonate release by the endomembrane Ca2+-ATPase inhibitor thapsigargin. Evidence for the involvement of a Ca2+-dependent route of priming in the production of lipid mediators by human polymorphonuclear leukocytes. Journal of Biological Chemistry. 1993;268(33):24751–24757. - PubMed
-
- Buczynski MW, Stephens DL, Bowers-Gentry RC, Grkovich A, Deems RA, Dennis EA. TLR-4 and sustained calcium agonists synergistically produce eicosanoids independent of protein synthesis in RAW264.7 cells. Journal of Biological Chemistry. 2007;282(31):22834–22847. - PubMed
-
- Lindner SC, Köhl U, Maier TJ, Steinhilber D, Sorg BL. TLR2 ligands augment cPLA2 α activity and lead to enhanced leukotriene release in human monocytes. Journal of Leukocyte Biology. 2009;86(2):389–399. - PubMed
-
- Akira S, Takeda K. Toll-like receptor signalling. Nature Reviews Immunology. 2004;4(7):499–511. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

