A critical tryptophan and Ca2+ in activation and catalysis of TPPI, the enzyme deficient in classic late-infantile neuronal ceroid lipofuscinosis
- PMID: 20689811
- PMCID: PMC2914745
- DOI: 10.1371/journal.pone.0011929
A critical tryptophan and Ca2+ in activation and catalysis of TPPI, the enzyme deficient in classic late-infantile neuronal ceroid lipofuscinosis
Abstract
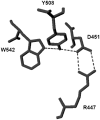
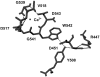
Background: Tripeptidyl aminopeptidase I (TPPI) is a crucial lysosomal enzyme that is deficient in the fatal neurodegenerative disorder called classic late-infantile neuronal ceroid lipofuscinosis (LINCL). It is involved in the catabolism of proteins in the lysosomes. Recent X-ray crystallographic studies have provided insights into the structural/functional aspects of TPPI catalysis, and indicated presence of an octahedrally coordinated Ca(2+).
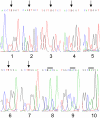
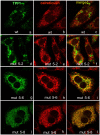
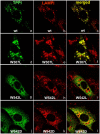
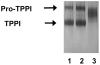
Methodology: Purified precursor and mature TPPI were used to study inhibition by NBS and EDTA using biochemical and immunological approaches. Site-directed mutagenesis with confocal imaging technique identified a critical W residue in TPPI activity, and the processing of precursor into mature enzyme.
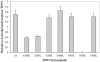
Principal findings: NBS is a potent inhibitor of the purified TPPI. In mammalian TPPI, W542 is critical for tripeptidyl peptidase activity as well as autocatalysis. Transfection studies have indicated that mutants of the TPPI that harbor residues other than W at position 542 have delayed processing, and are retained in the ER rather than transported to lysosomes. EDTA inhibits the autocatalytic processing of the precursor TPPI.
Conclusions/significance: We propose that W542 and Ca(2+) are critical for maintaining the proper tertiary structure of the precursor proprotein as well as the mature TPPI. Additionally, Ca(2+) is necessary for the autocatalytic processing of the precursor protein into the mature TPPI. We have identified NBS as a potent TPPI inhibitor, which led in delineating a critical role for W542 residue. Studies with such compounds will prove valuable in identifying the critical residues in the TPPI catalysis and its structure-function analysis.
Conflict of interest statement
Figures

References
-
- Junaid MA, Pullarkat RK. Biochemistry of neuronal ceroid lipofuscinosis. Adv Genet. 2001;45:93–106. - PubMed
-
- Golabek AA, Kida E. Tripeptidyl-peptidase I in health and disease. Biol Chem. 2006;387(8):1091–1099. - PubMed
-
- Zeman W, Donahue S, Dyken P, Green J. Leukodystrophies and poliodystrophies. In: Vinken PJ, Bruyn GW, editors. Handbook of Clinical Neurology. Amsterdam: Elsevier Science. 1970;Volume 10:588–679.
-
- Boustany R-M. Neurodystrophies and Neurolipidoses. In: Moser HW, editor. Handbook of Clinical Neurology. Amsterdam: Elsevier Science. 1996;Volume 22 (66):671–700.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

