Retinoic acids potentiate BMP9-induced osteogenic differentiation of mesenchymal progenitor cells
- PMID: 20689834
- PMCID: PMC2912873
- DOI: 10.1371/journal.pone.0011917
Retinoic acids potentiate BMP9-induced osteogenic differentiation of mesenchymal progenitor cells
Abstract
Background: As one of the least studied bone morphogenetic proteins (BMPs), BMP9 is one of the most osteogenic BMPs. Retinoic acid (RA) signaling is known to play an important role in development, differentiation and bone metabolism. In this study, we investigate the effect of RA signaling on BMP9-induced osteogenic differentiation of mesenchymal progenitor cells (MPCs).
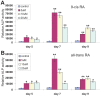
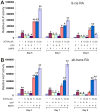
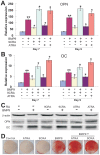
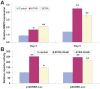
Methodology/principal findings: Both primary MPCs and MPC line are used for BMP9 and RA stimulation. Recombinant adenoviruses are used to deliver BMP9, RARalpha and RXRalpha into MPCs. The in vitro osteogenic differentiation is monitored by determining the early and late osteogenic markers and matrix mineralization. Mouse perinatal limb explants and in vivo MPC implantation experiments are carried out to assess bone formation. We find that both 9CRA and ATRA effectively induce early osteogenic marker, such as alkaline phosphatase (ALP), and late osteogenic markers, such as osteopontin (OPN) and osteocalcin (OC). BMP9-induced osteogenic differentiation and mineralization is synergistically enhanced by 9CRA and ATRA in vitro. 9CRA and ATRA are shown to induce BMP9 expression and activate BMPR Smad-mediated transcription activity. Using mouse perinatal limb explants, we find that BMP9 and RAs act together to promote the expansion of hypertrophic chondrocyte zone at growth plate. Progenitor cell implantation studies reveal that co-expression of BMP9 and RXRalpha or RARalpha significantly increases trabecular bone and osteoid matrix formation.
Conclusion/significance: Our results strongly suggest that retinoid signaling may synergize with BMP9 activity in promoting osteogenic differentiation of MPCs. This knowledge should expand our understanding about how BMP9 cross-talks with other signaling pathways. Furthermore, a combination of BMP9 and retinoic acid (or its agonists) may be explored as effective bone regeneration therapeutics to treat large segmental bony defects, non-union fracture, and/or osteoporotic fracture.
Conflict of interest statement
Figures
References
-
- Sacchetti B, Funari A, Michienzi S, Di Cesare S, Piersanti S, et al. Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell. 2007;131:324–336. - PubMed
-
- Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;276:71–74. - PubMed
-
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. - PubMed
-
- Aubin JE. Advances in the osteoblast lineage. Biochem Cell Biol. 1998;76:899–910. - PubMed
-
- Deng ZL, Sharff KA, Tang N, Song WX, Luo J, et al. Regulation of osteogenic differentiation during skeletal development. Front Biosci. 2008;13:2001–2021. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

