GIP increases human adipocyte LPL expression through CREB and TORC2-mediated trans-activation of the LPL gene
- PMID: 20693566
- PMCID: PMC2952555
- DOI: 10.1194/jlr.M006841
GIP increases human adipocyte LPL expression through CREB and TORC2-mediated trans-activation of the LPL gene
Abstract
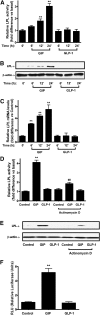
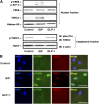
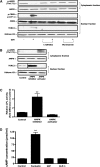
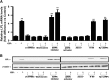
GIP (glucose-dependent insulinotropic polypeptide) is a gastrointestinal hormone that regulates pancreatic islet function. Additionally, emerging evidence suggests an important physiological role for GIP in the regulation of adipocyte metabolism. In previous studies on the lipogenic effects of GIP, it was shown to increase adipocyte lipoprotein lipase (LPL) activity in both differentiated 3T3-L1 cells and human adipocytes through a pathway involving activation of protein kinase B (PKB)/Akt. In the current study, we examined the effects of GIP on LPL gene expression. GIP in the presence of insulin increased LPL gene expression in human adipocytes and LPL promoter activity in GIP receptor-expressing HEK-293 cells, and both effects were greatly reduced by the transcription inhibitor actinomycin D. Subsequent studies established that GIP increased phosphorylation of Serine 133 in cAMP-response element binding protein (CREB) and the nuclear localization of cAMP-responsive CREB coactivator 2 (TORC2) through a pathway involving phosphatidylinositol 3-kinase (PI3-K), PKB, and AMP-activated protein kinase (AMPK). However, in the presence of insulin, GIP failed to activate the cAMP/PKA pathway. Knockdown of CREB and TORC2 using RNA interference reduced LPL expression, supporting a functional regulatory role. GIP-induced phospho-CREB and TORC2 were shown to bind to a cAMP-response element (-II) site in the human LPL promoter and GIP increased protein-protein interactions of these two factors. The lipogenic effects of GIP in the presence of insulin are therefore at least partially mediated by upregulation of adipocyte LPL gene transcription through a pathway involving PI3-K/PKB/AMPK-dependent CREB/TORC2 activation.
Figures



Similar articles
-
Glucose-dependent insulinotropic polypeptide-mediated up-regulation of beta-cell antiapoptotic Bcl-2 gene expression is coordinated by cyclic AMP (cAMP) response element binding protein (CREB) and cAMP-responsive CREB coactivator 2.Mol Cell Biol. 2008 Mar;28(5):1644-56. doi: 10.1128/MCB.00325-07. Epub 2007 Dec 17. Mol Cell Biol. 2008. PMID: 18086876 Free PMC article.
-
Resistin is a key mediator of glucose-dependent insulinotropic polypeptide (GIP) stimulation of lipoprotein lipase (LPL) activity in adipocytes.J Biol Chem. 2007 Nov 23;282(47):34139-47. doi: 10.1074/jbc.M704896200. Epub 2007 Sep 20. J Biol Chem. 2007. PMID: 17890220
-
Resistin knockout mice exhibit impaired adipocyte glucose-dependent insulinotropic polypeptide receptor (GIPR) expression.Diabetes. 2013 Feb;62(2):471-7. doi: 10.2337/db12-0257. Epub 2012 Sep 21. Diabetes. 2013. PMID: 23002036 Free PMC article.
-
Glucose-dependent insulinotropic polypeptide signaling in pancreatic β-cells and adipocytes.J Diabetes Investig. 2012 Mar 28;3(2):96-106. doi: 10.1111/j.2040-1124.2012.00196.x. J Diabetes Investig. 2012. PMID: 24843552 Free PMC article. Review.
-
Biological functions of CRTC2 and its role in metabolism-related diseases.J Cell Commun Signal. 2023 Sep;17(3):495-506. doi: 10.1007/s12079-023-00730-5. Epub 2023 Mar 1. J Cell Commun Signal. 2023. PMID: 36856929 Free PMC article. Review.
Cited by
-
Mechanisms underlying the onset of oral lipid-induced skeletal muscle insulin resistance in humans.Diabetes. 2013 Jul;62(7):2240-8. doi: 10.2337/db12-1179. Epub 2013 Mar 1. Diabetes. 2013. PMID: 23454694 Free PMC article. Clinical Trial.
-
Tirzepatide: A Systematic Update.Int J Mol Sci. 2022 Nov 23;23(23):14631. doi: 10.3390/ijms232314631. Int J Mol Sci. 2022. PMID: 36498958 Free PMC article. Review.
-
Intestinal lipid absorption and transport in type 2 diabetes.Diabetologia. 2022 Oct;65(10):1587-1600. doi: 10.1007/s00125-022-05765-8. Epub 2022 Jul 30. Diabetologia. 2022. PMID: 35908083 Review.
-
Pharmacological Advances in Incretin-Based Polyagonism: What We Know and What We Don't.Physiology (Bethesda). 2024 May 1;39(3):142-156. doi: 10.1152/physiol.00032.2023. Epub 2024 Feb 14. Physiology (Bethesda). 2024. PMID: 38353610 Free PMC article. Review.
-
Tirzepatide, GIP(1-42) and GIP(1-30) display unique signaling profiles at two common GIP receptor variants, E354 and Q354.Front Pharmacol. 2024 Oct 11;15:1463313. doi: 10.3389/fphar.2024.1463313. eCollection 2024. Front Pharmacol. 2024. PMID: 39464637 Free PMC article.
References
-
- Drucker D. J., Nauck M. A. 2006. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 368: 1696–1705. - PubMed
-
- McIntosh C. H. S., Widenmaier S., Kim S. J. 2009. Glucose-dependent insulinotropic polypeptide (Gastric Inhibitory Polypeptide; GIP). Vitam. Horm. 80: 409–471. - PubMed
-
- Davidson M. B., Bate G., Kirkpatrick P. 2005. Exenatide. Nat. Rev. Drug Discov. 4: 713–714. - PubMed
-
- Harder H., Nielsen L., Tu D. T. T., Astrup A. 2004. The effect of liraglutide, a long-acting glucagon-like peptide 1 derivative, on glycemic control, body composition, and 24-h energy expenditure in patients with type 2 diabetes. Diabetes Care. 27: 1915–1921. - PubMed
-
- DeFronzo R. A., Ratner R. E., Han J., Kim D. D., Fineman M. S., Baron A. D. 2005. Effects of exenatide (exendin-4) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care. 28: 1092–1100. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

