Nontoxic genetic engineering of mesenchymal stem cells using serum-compatible pullulan-spermine/DNA anioplexes
- PMID: 20698746
- PMCID: PMC3053036
- DOI: 10.1089/ten.TEC.2010.0120
Nontoxic genetic engineering of mesenchymal stem cells using serum-compatible pullulan-spermine/DNA anioplexes
Abstract
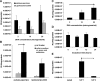
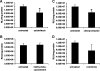
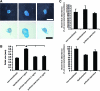
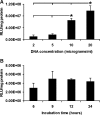
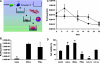
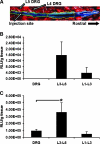
Genetic modification of stem cells could be applied to initiate/enhance their secretion of therapeutic molecules, alter their biological properties, or label them for in vivo tracking. We recently developed a negatively charged gene carrier ("anioplex") based on pullulan-spermine, a conjugate prepared from a natural polysaccharide and polyamine. In rat mesenchymal stem cells (MSCs), anioplex-derived reporter gene activity was comparable to or exceeded that obtained using a commercial cationic lipid reagent. Transfection in the growth medium with 15% serum and antibiotics was approximately sevenfold more effective than in serum-free conditions. Cytotoxicity was essentially indiscernible after 24 h of anioplex transfection with 20 μg/mL DNA, in contrast to cationic lipid transfection that resulted in 40%-60% death of target MSCs. Anioplex-derived reporter gene activity persisted throughout the entire 3-week study, with post-transfection MSCs appearing to maintain osteogenic, adipogenic, and chondrogenic multipotency. In particular, chondrogenic pellet formation of differentiating human MSCs was significantly inhibited after lipofection but not after aniofection, which further indicates the biological inertness of pullulan-spermine/DNA anioplexes. Collectively, these data introduce a straightforward technology for genetic engineering of adult stem/progenitor cells under physiological niche-like conditions. Moreover, reporter gene activity was observed in rat spinal cords after minimally invasive intrathecal implantation, suggesting effective engraftment of donor MSCs. It is therefore plausible that anioplex-transfected MSCs or other stem/progenitor cells with autologous potential could be applied to disorders such as neurotrauma or neuropathic pain that involve the spinal cord and brain.
Figures

Similar articles
-
Neuronal gene delivery by negatively charged pullulan-spermine/DNA anioplexes.Biomaterials. 2009 Mar;30(9):1815-26. doi: 10.1016/j.biomaterials.2008.12.032. Epub 2009 Jan 18. Biomaterials. 2009. PMID: 19152971
-
Preparation of cationized polysaccharides as gene transfection carrier for bone marrow-derived mesenchymal stem cells.J Biomater Sci Polym Ed. 2010;21(2):185-204. doi: 10.1163/156856209X415495. J Biomater Sci Polym Ed. 2010. PMID: 20092684
-
Reversal of tumor growth by gene modification of mesenchymal stem cells using spermine-pullulan/DNA nanoparticles.J Biomed Nanotechnol. 2014 Feb;10(2):299-308. doi: 10.1166/jbn.2014.1712. J Biomed Nanotechnol. 2014. PMID: 24738338
-
A reverse transfection technology to genetically engineer adult stem cells.Tissue Eng. 2007 Feb;13(2):245-51. doi: 10.1089/ten.2006.0185. Tissue Eng. 2007. PMID: 17518561
-
TGF-β1 regulates differentiation of bone marrow mesenchymal stem cells.Vitam Horm. 2011;87:127-41. doi: 10.1016/B978-0-12-386015-6.00042-1. Vitam Horm. 2011. PMID: 22127241 Review.
Cited by
-
Transplanting Mesenchymal Stem Cells for Treatment of Ischemic Stroke.Cell Transplant. 2018 Dec;27(12):1825-1834. doi: 10.1177/0963689718795424. Epub 2018 Sep 25. Cell Transplant. 2018. PMID: 30251564 Free PMC article. Review.
-
Osteocalcin expressing cells from tendon sheaths in mice contribute to tendon repair by activating Hedgehog signaling.Elife. 2017 Dec 15;6:e30474. doi: 10.7554/eLife.30474. Elife. 2017. PMID: 29244023 Free PMC article.
-
Gene Delivery Approaches for Mesenchymal Stem Cell Therapy: Strategies to Increase Efficiency and Specificity.Stem Cell Rev Rep. 2017 Dec;13(6):725-740. doi: 10.1007/s12015-017-9760-2. Stem Cell Rev Rep. 2017. PMID: 28815481 Review.
-
Adipose-derived stromal cells overexpressing vascular endothelial growth factor accelerate mouse excisional wound healing.Mol Ther. 2013 Feb;21(2):445-55. doi: 10.1038/mt.2012.234. Epub 2012 Nov 20. Mol Ther. 2013. PMID: 23164936 Free PMC article.
-
Direct Cell Reprogramming and Phenotypic Conversion: An Analysis of Experimental Attempts to Transform Astrocytes into Neurons in Adult Animals.Cells. 2023 Feb 14;12(4):618. doi: 10.3390/cells12040618. Cells. 2023. PMID: 36831283 Free PMC article. Review.
References
-
- Kumar S. Chanda D. Ponnazhagan S. Therapeutic potential of genetically modified mesenchymal stem cells. Gene Ther. 2008;15:711. - PubMed
-
- Jäderstad J. Jäderstad L.M. Li J. Chintawar S. Salto C. Pandolfo M. Ourednik V. Teng Y.D. Sidman R.L. Arenas E. Snyder E.Y. Herlenius E. Communication via gap junctions underlies early functional and beneficial interactions between grafted neural stem cells and the host. Proc Natl Acad Sci U S A. 2010;107:5184. - PMC - PubMed
-
- Giudice A. Trounson A. Genetic modification of human embryonic stem cells for derivation of target cells. Cell Stem Cell. 2008;2:422. - PubMed
-
- Tey S.K. Brenner M.K. The continuing contribution of gene marking to cell and gene therapy. Mol Ther. 2007;15:666. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

