A family of intermediate filament-like proteins is sequentially assembled into the cytoskeleton of Toxoplasma gondii
- PMID: 20698859
- PMCID: PMC3005026
- DOI: 10.1111/j.1462-5822.2010.01514.x
A family of intermediate filament-like proteins is sequentially assembled into the cytoskeleton of Toxoplasma gondii
Abstract
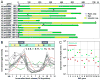
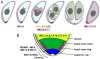
The intracellular protozoan parasite Toxoplasma gondii divides by a unique process of internal budding that involves the assembly of two daughter cells within the mother. The cytoskeleton of Toxoplasma, which is composed of microtubules associated with an inner membrane complex (IMC), has an important role in this process. The IMC, which is directly under the plasma membrane, contains a set of flattened membranous sacs lined on the cytoplasmic side by a network of filamentous proteins. This network contains a family of intermediate filament-like proteins or IMC proteins. In order to elucidate the division process, we have characterized a 14-member subfamily of Toxoplasma IMC proteins that share a repeat motif found in proteins associated with the cortical alveoli in all alveolates. By creating fluorescent protein fusion reporters for the family members we determined the spatiotemporal patterns of all 14 IMC proteins through tachyzoite development. This revealed several distinct distribution patterns and some provide the basis for novel structural models such as the assembly of certain family members into the basal complex. Furthermore we identified IMC15 as an early marker of budding and, lastly, the dynamic patterns observed throughout cytokinesis provide a timeline for daughter parasite development and division.
© 2010 Blackwell Publishing Ltd.
Figures





References
-
- Alexandrov A, Vignali M, LaCount DJ, Quartley E, de Vries C, De Rosa D, et al. A facile method for high-throughput co-expression of protein pairs. Mol Cell Proteomics. 2004;3:934–938. - PubMed
-
- Ferguson DJ, Cesbron-Delauw MF, Dubremetz JF, Sibley LD, Joiner KA, Wright S. The expression and distribution of dense granule proteins in the enteric (Coccidian) forms of Toxoplasma gondii in the small intestine of the cat. Exp Parasitol. 1999;91:203–211. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

