Body weight control by a high-carbohydrate/low-fat diet slows the progression of diabetic kidney damage in an obese, hypertensive, type 2 diabetic rat model
- PMID: 20700413
- PMCID: PMC2911582
- DOI: 10.1155/2010/136502
Body weight control by a high-carbohydrate/low-fat diet slows the progression of diabetic kidney damage in an obese, hypertensive, type 2 diabetic rat model
Abstract
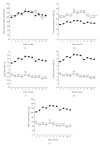
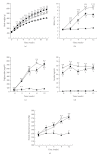
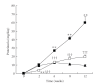
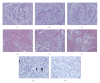
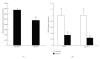
Obesity is one of several factors implicated in the genesis of diabetic nephropathy (DN). Obese, hypertensive, type 2 diabetic rats SHR/NDmcr-cp were given, for 12 weeks, either a normal, middle-carbohydrate/middle-fat diet (MC/MF group) or a high-carbohydrate/low-fat diet (HC/LF group). Daily caloric intake was the same in both groups. Nevertheless, the HC/LF group gained less weight. Despite equivalent degrees of hypertension, hyperglycemia, hyperlipidemia, hyperinsulinemia, and even a poorer glycemic control, the HC/LF group had less severe renal histological abnormalities and a reduced intrarenal advanced glycation and oxidative stress. Mediators of the renoprotection, specifically linked to obesity and body weight control, include a reduced renal inflammation and TGF-beta expression, together with an enhanced level of adiponectin. Altogether, these data identify a specific role of body weight control by a high-carbohydrate/low-fat diet in the progression of DN. Body weight control thus impacts on local intrarenal advanced glycation and oxidative stress through inflammation and adiponectin levels.
Figures
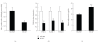
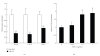
Similar articles
-
Tempol or candesartan prevents high-fat diet-induced hypertension and renal damage in spontaneously hypertensive rats.Nephrol Dial Transplant. 2010 Feb;25(2):389-99. doi: 10.1093/ndt/gfp472. Epub 2009 Sep 11. Nephrol Dial Transplant. 2010. PMID: 19749146
-
In a type 2 diabetic nephropathy rat model, the improvement of obesity by a low calorie diet reduces oxidative/carbonyl stress and prevents diabetic nephropathy.Nephrol Dial Transplant. 2005 Dec;20(12):2661-9. doi: 10.1093/ndt/gfi096. Epub 2005 Sep 27. Nephrol Dial Transplant. 2005. PMID: 16188903
-
Inhibition of advanced glycation end products (AGEs): an implicit goal in clinical medicine for the treatment of diabetic nephropathy?Diabetes Res Clin Pract. 2008 Nov 13;82 Suppl 1:S25-9. doi: 10.1016/j.diabres.2008.09.012. Epub 2008 Oct 26. Diabetes Res Clin Pract. 2008. PMID: 18954918
-
Cobalt ameliorates renal injury in an obese, hypertensive type 2 diabetes rat model.Nephrol Dial Transplant. 2008 Apr;23(4):1166-72. doi: 10.1093/ndt/gfm715. Epub 2007 Oct 28. Nephrol Dial Transplant. 2008. PMID: 17967803
-
Effects of macronutrient intake in obesity: a meta-analysis of low-carbohydrate and low-fat diets on markers of the metabolic syndrome.Nutr Rev. 2021 Mar 9;79(4):429-444. doi: 10.1093/nutrit/nuaa044. Nutr Rev. 2021. PMID: 32885229 Free PMC article. Review.
Cited by
-
Unilateral nephrectomized SHR/NDmcr-cp rat shows a progressive decline of glomerular filtration with tubular interstitial lesions.Physiol Res. 2023 Apr 30;72(2):209-220. doi: 10.33549/physiolres.934969. Physiol Res. 2023. PMID: 37159855 Free PMC article.
-
Relationship between Glycemic Load and Blood Lipid Level in Hospitalized Adult Chinese.Iran J Public Health. 2015 Mar;44(3):318-24. Iran J Public Health. 2015. PMID: 25905074 Free PMC article.
-
Aldosterone antagonists in monotherapy are protective against streptozotocin-induced diabetic nephropathy in rats.PLoS One. 2012;7(6):e39938. doi: 10.1371/journal.pone.0039938. Epub 2012 Jun 28. PLoS One. 2012. PMID: 22761931 Free PMC article.
-
High-Carbohydrate Diet Consumption Poses a More Severe Liver Cholesterol Deposition than a High-Fat and High-Calorie Diet in Mice.Int J Mol Sci. 2023 Sep 28;24(19):14700. doi: 10.3390/ijms241914700. Int J Mol Sci. 2023. PMID: 37834148 Free PMC article.
References
-
- Sarafidis PA, Ruilope LM. Insulin resistance, hyperinsulinemia, and renal injury: mechanisms and implications. American Journal of Nephrology. 2006;26(3):232–244. - PubMed
-
- Trovati M, Cavalot F. Optimization of hypolipidemic and antiplatelet treatment in the diabetic patient with renal diseas. Journal of the American Society of Nephrology. 2004;15(supplement 1):S12–S20. - PubMed
-
- Sarafidis PA, Bakris GL. Protection of the kidney by thiazolidinediones: an assessment from bench to bedside. Kidney International. 2006;70(7):1223–1233. - PubMed
-
- Remuzzi G, Macia M, Ruggenenti P. Prevention and treatment of diabetic renal disease in type 2 diabetes: the BENEDICT study. Journal of the American Society of Nephrology. 2006;17(supplement 2):S90–S97. - PubMed
-
- Fioretto P, Bruseghin M, Berto I, Gallina P, Manzato E, Mussap M. Renal protection in diabetes: role of glycemic control. Journal of the American Society of Nephrology. 2006;17(supplement 2):S86–S89. - PubMed
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

