Stereoselective hydrolysis of organophosphate nerve agents by the bacterial phosphotriesterase
- PMID: 20701311
- PMCID: PMC2945820
- DOI: 10.1021/bi101056m
Stereoselective hydrolysis of organophosphate nerve agents by the bacterial phosphotriesterase
Abstract
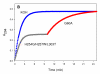
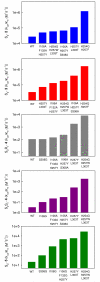
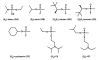
Organophosphorus compounds include many synthetic, neurotoxic substances that are commonly used as insecticides. The toxicity of these compounds is due to their ability to inhibit the enzyme acetylcholine esterase. Some of the most toxic organophosphates have been adapted for use as chemical warfare agents; the most well-known are GA, GB, GD, GF, VX, and VR. All of these compounds contain a chiral phosphorus center, with the S(P) enantiomers being significantly more toxic than the R(P) enantiomers. Phosphotriesterase (PTE) is an enzyme capable of detoxifying these agents, but the stereochemical preference of the wild-type enzyme is for the R(P) enantiomers. A series of enantiomerically pure chiral nerve agent analogues containing the relevant phosphoryl centers found in GB, GD, GF, VX, and VR has been developed. Wild-type and mutant forms of PTE have been tested for their ability to hydrolyze this series of compounds. Mutant forms of PTE with significantly enhanced, as well as relaxed or reversed, stereoselectivity have been identified. A number of variants exhibited dramatically improved kinetic constants for the catalytic hydrolysis of the more toxic S(P) enantiomers. Improvements of up to 3 orders of magnitude relative to the value of the wild-type enzyme were observed. Some of these mutants were tested against racemic mixtures of GB and GD. The kinetic constants obtained with the chiral nerve agent analogues accurately predict the improved activity and stereoselectivity against the authentic nerve agents used in this study.
Figures






References
-
- Ecobichon DJ. Casarett and Doull’s Toxicology: The basic science of poisons. 6th ed McGraw-Hill; New York: 2001. pp. 763–810.
-
- Benschop HP, Dejong LPA. Nerve agent stereoisomers - analysis, isolation, and toxicology. Accounts of Chemical Research. 1988;21:368–374.
-
- Worek F, Szinicz L, Eyer P, Thiermann H. Evaluation of oxime efficacy in nerve agent poisoning: Development of a kinetic-based dynamic model. Toxicology and Applied Pharmacology. 2005;209:193–202. - PubMed
-
- Josse D, Xie W, Renault F, Rochu F, Schopfer LM, Masson P, Lockridge O. Identification of Residues Essential for Human Paraoxonase (PON1) Arylesterase/Organophosphatase Activities. Biochemistry. 1999;38:2816–2825. - PubMed
-
- Hartleib J, Ruterjans H. High-yield expression, purification, and characterization of the recombinant diisopropylfluorophosphatase from Loligo vulgaris. Protein Expression and Purification. 2001;21:210–219. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

