Temporally extended dopamine responses to perceptually demanding reward-predictive stimuli
- PMID: 20702700
- PMCID: PMC3297489
- DOI: 10.1523/JNEUROSCI.4828-09.2010
Temporally extended dopamine responses to perceptually demanding reward-predictive stimuli
Abstract
Midbrain dopamine neurons respond to reward-predictive stimuli. In the natural environment reward-predictive stimuli are often perceptually complicated. Thus, to discriminate one stimulus from another, elaborate sensory processing is necessary. Given that previous studies have used simpler types of reward-predictive stimuli, it has yet to be clear whether and, if so, how dopamine neurons obtain reward information from perceptually complicated stimuli. To investigate this, we recorded the activities of monkey dopamine neurons while they were performing discrimination between two coherent motion directions in random-dot motion stimuli. These coherent directions were paired with different magnitudes of reward. We found that dopamine neurons showed reward-predictive responses to random-dot motion stimuli. Moreover, dopamine neurons showed temporally extended activity correlated with changes in reward prediction (i.e., reward prediction error) from coarse to fine scales between initial motion detection and subsequent motion discrimination phases. Noticeably, dopamine reward-predictive responses became differential in a later phase than previously reported. This response pattern was consistent with the time course of processing required for the estimation of expected reward value that parallels the motion direction discrimination processing. The results demonstrate that dopamine neurons are able to reflect the reward value of perceptually complicated stimuli, and suggest that dopamine neurons use the moment-to-moment reward prediction associated with environmental stimuli to compute a reward prediction error.
Figures


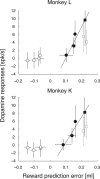
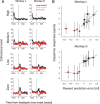

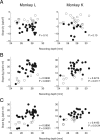
References
-
- Ditterich J, Mazurek ME, Shadlen MN. Microstimulation of visual cortex affects the speed of perceptual decisions. Nat Neurosci. 2003;6:891–898. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
