Application of phase-trafficking methods to natural products research
- PMID: 20704309
- PMCID: PMC2945427
- DOI: 10.1021/np100465h
Application of phase-trafficking methods to natural products research
Erratum in
- J Nat Prod. 2011 Apr 25;74(4):916
Abstract
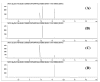
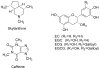
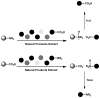
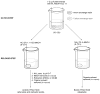
A novel simultaneous phase-trafficking approach using spatially separated solid-supported reagents for rapid separation of neutral, basic, and acidic compounds from organic plant extracts with minimum labor is reported. Acidic and basic ion-exchange resins were physically separated into individual sacks ("tea bags") for trapping basic and acidic compounds, respectively, leaving behind in solution neutral components of the natural mixtures. Trapped compounds were then recovered from solid phase by appropriate suspension in acidic or basic solutions. The feasibility of the proposed separation protocol was demonstrated and optimized with an "artificial mixture" of model compounds. In addition, the utility of this methodology was illustrated with the successful separation of the alkaloid skytanthine from Skytanthus acutus Meyen and the main catechins and caffeine from Camellia sinensis L. (Kuntze). This novel approach offers multiple advantages over traditional extraction methods, as it is not labor intensive, makes use of only small quantities of solvents, produces fractions in adequate quantities for biological assays, and can be easily adapted to field conditions for bioprospecting activities.
Figures


Similar articles
-
Molecular interactions between caffeine and catechins in green tea.J Agric Food Chem. 2014 Jul 16;62(28):6777-83. doi: 10.1021/jf5011287. Epub 2014 Jul 8. J Agric Food Chem. 2014. PMID: 24980214
-
[Determination of catechins and caffeine in tea and tea beverages by high-performance liquid chromatography].Wei Sheng Yan Jiu. 2005 Mar;34(2):187-90. Wei Sheng Yan Jiu. 2005. PMID: 15952660 Chinese.
-
Analysis of some selected catechins and caffeine in green tea by high performance liquid chromatography.Food Chem. 2012 Oct 15;134(4):2268-75. doi: 10.1016/j.foodchem.2012.03.039. Epub 2012 Mar 28. Food Chem. 2012. PMID: 23442685
-
Characterizing the cultivar-specific mechanisms underlying the accumulation of quality-related metabolites in specific Chinese tea (Camellia sinensis) germplasms to diversify tea products.Food Res Int. 2022 Nov;161:111824. doi: 10.1016/j.foodres.2022.111824. Epub 2022 Aug 24. Food Res Int. 2022. PMID: 36192959 Review.
-
Green preparation, safety control and intelligent processing of high-quality tea extract.Crit Rev Food Sci Nutr. 2024 Nov;64(31):11468-11492. doi: 10.1080/10408398.2023.2239348. Epub 2023 Jul 26. Crit Rev Food Sci Nutr. 2024. PMID: 37493455 Review.
Cited by
-
Siloxyl ether functionalized resins for chemoselective enrichment of carboxylic acids.Org Lett. 2011 Oct 21;13(20):5652-5. doi: 10.1021/ol202376m. Epub 2011 Sep 29. Org Lett. 2011. PMID: 21957886 Free PMC article.
-
Chemoselective enrichment for natural products discovery.Chem Sci. 2011 Apr 1;2(4):760-764. doi: 10.1039/C0SC00620C. Epub 2011 Jan 28. Chem Sci. 2011. PMID: 24926410 Free PMC article.
-
Identification of unprecedented purine-containing compounds, the zingerines, from ginger rhizomes (Zingiber officinale Roscoe) using a phase-trafficking approach.Phytochemistry. 2011 Jun;72(9):935-41. doi: 10.1016/j.phytochem.2011.03.007. Epub 2011 Apr 15. Phytochemistry. 2011. PMID: 21497863 Free PMC article.
References
-
- Merrifield RB. J Am Chem Soc. 1963;85:2149–2154.
-
- Cano M, Balasubramanian S. Drugs Future. 2003;28:659–678.
-
- Alexandratos SD. Ind Eng Chem Res. 2009;48:388–398.
-
- Solinas A, Taddei M. Synthesis-Stuttgart. 2007;16:2409–2453.
-
- Kennedy JP, Williams L, Bridges TM, Daniels RN, Weaver D, Lindsley CW. J Comb Chem. 2008;10:345–354. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

