Characterization of azido-NAD+ to assess its potential as a two-dimensional infrared probe of enzyme dynamics
- PMID: 20705046
- PMCID: PMC2963086
- DOI: 10.1016/j.ab.2010.08.008
Characterization of azido-NAD+ to assess its potential as a two-dimensional infrared probe of enzyme dynamics
Abstract
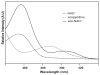
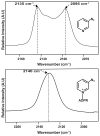
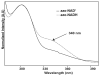
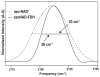
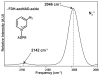
Enzyme active-site dynamics at femtosecond to picosecond time scales are of great biochemical importance, but remain relatively unexplored due to the lack of appropriate analytical methods. Two-dimensional infrared (2D IR) spectroscopy is one of the few methods that can examine chemical biological motions at this time scale, but all the IR probes used so far were specific to a few unique enzymes. The lack of IR probes of broader specificity is a major limitation to further 2D IR studies of enzyme dynamics. Here we describe the synthesis of a general IR probe for nicotinamide-dependent enzymes. This azido analog of the ubiquitous cofactor nicotinamide adenine dinucleotide is found to be stable and bind to several dehydrogenases with dissociation constants similar to that for the native cofactor. The infrared absorption spectra of this probe bound to several enzymes indicate that it has significant potential as a 2D IR probe to investigate femtosecond dynamics of nicotinamide-dependent enzymes.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Relationship of femtosecond-picosecond dynamics to enzyme-catalyzed H-transfer.Top Curr Chem. 2013;337:1-39. doi: 10.1007/128_2012_407. Top Curr Chem. 2013. PMID: 23539379 Free PMC article. Review.
-
Two-dimensional infrared spectroscopy of azido-nicotinamide adenine dinucleotide in water.J Chem Phys. 2011 Aug 7;135(5):055106. doi: 10.1063/1.3623418. J Chem Phys. 2011. PMID: 21823737 Free PMC article.
-
3-picolyl azide adenine dinucleotide as a probe of femtosecond to picosecond enzyme dynamics.J Phys Chem B. 2012 Jan 12;116(1):542-8. doi: 10.1021/jp208677u. Epub 2011 Dec 15. J Phys Chem B. 2012. PMID: 22126535 Free PMC article.
-
A Genetically Encoded Two-Dimensional Infrared Probe for Enzyme Active-Site Dynamics.Angew Chem Int Ed Engl. 2021 May 10;60(20):11143-11147. doi: 10.1002/anie.202016880. Epub 2021 Apr 7. Angew Chem Int Ed Engl. 2021. PMID: 33644946
-
Amide I two-dimensional infrared spectroscopy of proteins.Acc Chem Res. 2008 Mar;41(3):432-41. doi: 10.1021/ar700188n. Epub 2008 Feb 21. Acc Chem Res. 2008. PMID: 18288813 Review.
Cited by
-
Relationship of femtosecond-picosecond dynamics to enzyme-catalyzed H-transfer.Top Curr Chem. 2013;337:1-39. doi: 10.1007/128_2012_407. Top Curr Chem. 2013. PMID: 23539379 Free PMC article. Review.
-
Two-dimensional infrared spectroscopy of azido-nicotinamide adenine dinucleotide in water.J Chem Phys. 2011 Aug 7;135(5):055106. doi: 10.1063/1.3623418. J Chem Phys. 2011. PMID: 21823737 Free PMC article.
-
3-picolyl azide adenine dinucleotide as a probe of femtosecond to picosecond enzyme dynamics.J Phys Chem B. 2012 Jan 12;116(1):542-8. doi: 10.1021/jp208677u. Epub 2011 Dec 15. J Phys Chem B. 2012. PMID: 22126535 Free PMC article.
-
Synthesis of radiolabeled nicotinamide cofactors from labeled pyridines: versatile probes for enzyme kinetics.Anal Biochem. 2012 Nov 15;430(2):123-9. doi: 10.1016/j.ab.2012.08.012. Epub 2012 Aug 23. Anal Biochem. 2012. PMID: 22922383 Free PMC article.
-
Site-Specific Spectroscopic Reporters of the Local Electric Field, Hydration, Structure, and Dynamics of Biomolecules.J Phys Chem Lett. 2011 Sep 23;2:2598-2609. doi: 10.1021/jz201161b. J Phys Chem Lett. 2011. PMID: 22003429 Free PMC article.
References
-
- Finkelstein IJ, Zheng J, Ishikawa H, Kim S, Kwak K, Fayer MD. Probing dynamics of complex molecular systems with ultrafast 2D IR vibrational echo spectroscopy. Phys. Chem. Chem. Phys. 2007;9:1533–1549. - PubMed
-
- Boehr DD, McElheny D, Dyson HJ, Wright PE. The dynamic energy landscape of dihydrofolate reductase catalysis. Science. 2006;313:1638–1642. - PubMed
-
- Nagel ZD, Klinman JP. Tunneling and dynamics in enzymatic hydride transfer. Chem. Rev. 2006;106:3095–3118. - PubMed
-
- Kohen A. Kinetic isotope effects as probes for hydrogen tunneling, coupled motion and dynamics contributions to enzyme catalysis. Prog. React. Kinet. Mech. 2003;28:119–156.
-
- Kohen A. Current issues in enzymatic hydrogen transfer from carbon: tunneling and coupled motion from kinetic isotope effect studies. Hydrogen-Transfer React. 2007;4:1311–1340.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

