Pathogenesis of fasting and postprandial hyperglycemia in type 2 diabetes: implications for therapy
- PMID: 20705776
- PMCID: PMC2963523
- DOI: 10.2337/db10-1032
Pathogenesis of fasting and postprandial hyperglycemia in type 2 diabetes: implications for therapy
Abstract
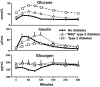
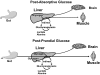
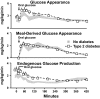
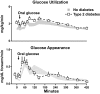
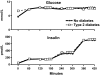
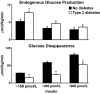
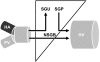
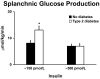
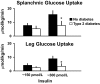
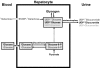
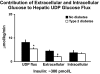
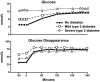
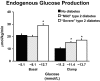
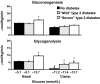
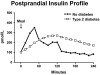
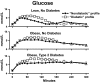
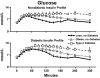
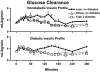
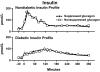
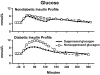
The objective of this research is to gain a greater understanding of the cause of fasting and postprandial hyperglycemia in people with type 2 diabetes. Endogenous glucose production is excessive before eating and fails to appropriately suppress after eating in people with type 2 diabetes. This is due in part to impaired insulin-induced suppression of endogenous glucose production, which is observed early in the evolution of type 2 diabetes. Increased rates of gluconeogenesis and perhaps glycogenolysis contribute to hepatic insulin resistance. Insulin-induced stimulation of hepatic glucose uptake and hepatic glycogen synthesis are reduced in people with type 2 diabetes primarily due to decreased uptake of extracellular glucose presumably because of inadequate activation of hepatic glucokinase. Delayed insulin secretion results in higher peak glucose concentrations particularly when suppression of glucagon is impaired, whereas insulin resistance prolongs the duration of hyperglycemia, which can be marked when both hepatic and extra-hepatic insulin resistance are present. The premise of these studies, as well as those performed by many other investigators, is that an understanding of the pathogenesis of type 2 diabetes will enable the development of targeted therapies that are directed toward correcting specific metabolic defects in a given individual. I, as well as many other investigators, believe that such therapies are likely to be more effective and to have a lower risk than would occur if everyone were treated the same regardless of the underlying cause of their hyperglycemia. While we do not yet have sufficient knowledge to truly individualize therapy, in my opinion this approach will be the norm in the not too distant future.
Figures



References
-
- Butler PC, Rizza RA. Contribution to postprandial hyperglycemia and effect on initial splanchnic glucose clearance of hepatic glucose cycling in glucose-intolerant or NIDDM patients. Diabetes 1991;40:73–81 - PubMed
-
- Reaven GM, Chen Y-DI, Coulston AM, Greenfield MS, Hollenbeck C, Lardinois C, Liu G, Schwartz H. Insulin secretion and action in noninsulin-dependent diabetes mellitus. Am J Med 1983;30:85–93 - PubMed
-
- Müller WA, Faloona GR, Aguilar-Parada E, Unger RH. Abnormal alpha-cell function in diabetes: response to carbohydrate and protein ingestion. N Engl J Med 1970;283:109–115 - PubMed
-
- Gerich JE, Lorenzi M, Karam JH, Schneider V, Forsham PH. Abnormal pancreatic glucagon secretion and postprandial hyperglycemia in diabetes mellitus. JAMA 1975;234:159–165 - PubMed
-
- Perley M, Kipnis DM. Plasma insulin responses to glucose and tolbutamide of normal weight and obese diabetic and nondiabetic subjects. Diabetes 1966;15:867–874 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

