Adiponectin-mediated heme oxygenase-1 induction protects against iron-induced liver injury via a PPARα dependent mechanism
- PMID: 20709802
- PMCID: PMC2947267
- DOI: 10.2353/ajpath.2010.090789
Adiponectin-mediated heme oxygenase-1 induction protects against iron-induced liver injury via a PPARα dependent mechanism
Abstract
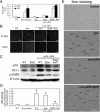
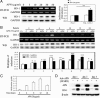
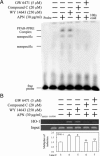
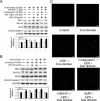
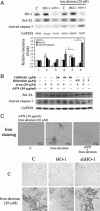
Protective effects of adiponectin (APN; an adipocytokine) were shown against various oxidative challenges; however, its therapeutic implications and the mechanisms underlying hepatic iron overload remain unclear. Herein, we show that the deleterious effects of iron dextran on liver function and iron deposition were significantly reversed by adiponectin gene therapy, which was accompanied by AMP-activated protein kinase (AMPK) phosphorylation and heme oxygenase (HO)-1 induction. Furthermore, AMPK-mediated peroxisome proliferator-activated receptor-α (PPARα) activation by APN was ascribable to HO-1 induction. Additionally, we revealed direct transcriptional regulation of HO-1 by the binding of PPARα to a PPAR-responsive element (PPRE) by various experimental assessments. Interestingly, overexpression of HO-1 in hepatocytes mimicked the protective effect of APN in attenuating iron-mediated injury, whereas it was abolished by SnPP and small interfering HO-1. Furthermore, bilirubin, the end-product of the HO-1 reaction, but not CO, protected hepatocytes from iron dextran-mediated caspase activation. Herein, we demonstrate a novel functional PPRE in the promoter regions of HO-1, and APN-mediated HO-1 induction elicited an antiapoptotic effect and a decrease in iron deposition in hepatocytes subjected to iron challenge.
Figures




References
-
- Rabin KR, Kamari Y, Avni I, Grossman E, Sharabi Y. Adiponectin: linking the metabolic syndrome to its cardiovascular consequences. Expert Rev Cardiovasc Ther. 2005;3:465–471. - PubMed
-
- Wulster-Radcliffe MC, Ajuwon KM, Wang J, Christian JA, Spurlock ME. Adiponectin differentially regulates cytokines in porcine macrophages. Biochem Biophys Res Commun. 2004;316:924–929. - PubMed
-
- Yokota T, Oritani K, Takahashi I, Ishikawa J, Matsuyama A, Ouchi N, Kihara S, Funahashi T, Tenner AJ, Tomiyama Y, Matsuzawa Y. Adiponectin, a new member of the family of soluble defense collagens, negatively regulates the growth of myelomonocytic progenitors and the functions of macrophages. Blood. 2000;96:1723–1732. - PubMed
-
- Yuhki K, Kawabe J, Ushikubi F. Fat, keeping the heart healthy? Nat Med. 2005;11:1048–1049. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

