The microrheology of sickle hemoglobin gels
- PMID: 20712998
- PMCID: PMC2920633
- DOI: 10.1016/j.bpj.2010.04.079
The microrheology of sickle hemoglobin gels
Abstract
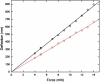
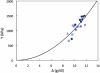
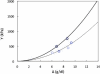
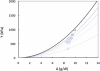
Sickle cell disease is a rheological disease, yet no quantitative rheological data exist on microscopic samples at physiological concentrations. We have developed a novel method for measuring the microrheology of sickle hemoglobin gels, based on magnetically driven compression of 5- to 8-microm-thick emulsions containing hemoglobin droplets approximately 80 microm in diameter. Using our method, by observing the expansion of the droplet area as the emulsion is compressed, we were able to resolve changes in thickness of a few nanometers with temporal resolution of milliseconds. Gels were formed at various initial concentrations and temperatures and with different internal domain structure. All behaved as Hookean springs with Young's modulus from 300 to 1500 kPa for gels with polymerized hemoglobin concentration from 6 g/dl to 12 g/dl. For uniform, multidomain gels, Young's modulus mainly depended on the terminal concentration of the gel rather than the conditions of formation. A simple model reproduced the quadratic dependence of the Young's modulus on the concentration of polymerized hemoglobin. Partially desaturated samples also displayed quadratic concentration dependence but with a smaller proportionality coefficient, as did samples that were desaturated in steps; such samples were significantly less rigid than gels formed all at once. The magnitude of the Young's modulus provides quantitative support for the dominant models of sickle pathophysiology.
2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Micromechanics of isolated sickle cell hemoglobin fibers: bending moduli and persistence lengths.J Mol Biol. 2002 Jan 25;315(4):601-12. doi: 10.1006/jmbi.2001.5130. J Mol Biol. 2002. PMID: 11812133
-
Depth-Dependent Out-of-Plane Young's Modulus of the Human Cornea.Curr Eye Res. 2018 May;43(5):595-604. doi: 10.1080/02713683.2017.1411951. Epub 2017 Dec 28. Curr Eye Res. 2018. PMID: 29283675
-
Exploring the mechanical properties of nanometer-thick elastic films through micro-drop impinging on large-area suspended graphene.Nanoscale. 2021 Dec 23;14(1):42-48. doi: 10.1039/d1nr05918a. Nanoscale. 2021. PMID: 34816842
-
Effect of other hemoglobins on gelation of sickle cell hemoglobin.Tex Rep Biol Med. 1980-1981;40:233-50. Tex Rep Biol Med. 1980. PMID: 6172866 Review. No abstract available.
-
Hemoglobin S gelation and sickle cell disease.Blood. 1987 Nov;70(5):1245-66. Blood. 1987. PMID: 3311198 Review. No abstract available.
Cited by
-
Ratchets, red cells, and metastability.Biophys Rev. 2013 Jun;5(2):217-224. doi: 10.1007/s12551-013-0117-z. Epub 2013 Apr 18. Biophys Rev. 2013. PMID: 28510163 Free PMC article. Review.
-
Multifunctional magnetic rotator for micro and nanorheological studies.Rev Sci Instrum. 2012 Jun;83(6):065110. doi: 10.1063/1.4729795. Rev Sci Instrum. 2012. PMID: 22755665 Free PMC article.
-
The physical foundation of vasoocclusion in sickle cell disease.Biophys J. 2012 Oct 17;103(8):L38-40. doi: 10.1016/j.bpj.2012.09.003. Epub 2012 Oct 16. Biophys J. 2012. PMID: 23083726 Free PMC article.
-
Novel single-cell measurements suggest irreversibly sickled cells are neither dense nor dehydrated.Biophys J. 2024 Nov 5;123(21):3750-3758. doi: 10.1016/j.bpj.2024.09.024. Epub 2024 Sep 26. Biophys J. 2024. PMID: 39327732
References
-
- Briehl R.W. Solid-like behaviour of unsheared sickle haemoglobin gels and the effects of shear. Nature. 1980;288:622–624. - PubMed
-
- Gabriel D.A., Smith L.A., Johnson C.S., Jr. Elastic properties of deoxy hemoglobin S (deoxy-HbS) gels. Arch. Biochem. Biophys. 1981;211:774–776. - PubMed
-
- Danish E.H., Harris J.W., Krieger I.M. Rheologic behavior of deoxyhemoglobin S gels. J. Mol. Biol. 1987;196:421–431. - PubMed
-
- Wang J.C., Turner M.S., Briehl R.W. Micromechanics of isolated sickle cell hemoglobin fibers: bending moduli and persistence lengths. J. Mol. Biol. 2002;315:601–612. - PubMed
-
- Turner M.S., Briehl R.W., Josephs R. Anisotropy in sickle hemoglobin fibers from variations in bending and twist. J. Mol. Biol. 2006;357:1422–1427. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

