The role of beta-arrestin2 in the severity of antinociceptive tolerance and physical dependence induced by different opioid pain therapeutics
- PMID: 20713067
- PMCID: PMC2981657
- DOI: 10.1016/j.neuropharm.2010.08.003
The role of beta-arrestin2 in the severity of antinociceptive tolerance and physical dependence induced by different opioid pain therapeutics
Abstract
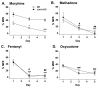
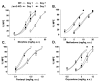
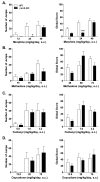
Ligands acting at the same receptor can differentially activate distinct signal transduction pathways, which in turn, can have diverse functional consequences. Further, receptors expressed in different tissues may utilize intracellular signaling proteins in response to a ligand differently as well. The mu opioid receptor (MOR), which mediates many of the pharmacological actions of opiate therapeutics, is also subject to differential signaling in response to diverse agonists. To study the effect of diverse agonists on MOR signaling, we examined the effects of chronic opiate treatment on two distinct physiological endpoints, antinociceptive tolerance and physical dependence, in mice lacking the intracellular regulatory molecule, βarrestin2. While βarrestin2 knockout (βarr2-KO) mice do not become tolerant to the antinociceptive effects of chronic morphine in a hot plate test, tolerance develops to the same degree in both wild type and βarr2-KO mice following chronic infusion with methadone, fentanyl, and oxycodone. Studies here also assess the severity of withdrawal signs precipitated by naloxone following chronic infusions at three different doses of each opiate agonist. While there are no differences in withdrawal responses between genotypes at the highest dose of morphine tested (48 mg/kg/day), the βarr2-KO mice display several less severe withdrawal responses when the infusion dose is lowered (12 or 24 mg/kg/day). Chronic infusion of methadone, fentanyl, and oxycodone all lead to equivalent naloxone-precipitated withdrawal responses in both genotypes at all doses tested. These results lend further evidence that distinct agonists can differentially impact on opioid-mediated responses in vivo in a βarrestin2-dependent manner.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Figures
Similar articles
-
Differential mechanisms of morphine antinociceptive tolerance revealed in (beta)arrestin-2 knock-out mice.J Neurosci. 2002 Dec 1;22(23):10494-500. doi: 10.1523/JNEUROSCI.22-23-10494.2002. J Neurosci. 2002. PMID: 12451149 Free PMC article.
-
Activation of delta-opioid receptor contributes to the antinociceptive effect of oxycodone in mice.Pharmacol Res. 2016 Sep;111:867-876. doi: 10.1016/j.phrs.2016.05.034. Epub 2016 Aug 2. Pharmacol Res. 2016. PMID: 27496654
-
Opioid agonist efficacy predicts the magnitude of tolerance and the regulation of mu-opioid receptors and dynamin-2.Eur J Pharmacol. 2007 Jun 1;563(1-3):92-101. doi: 10.1016/j.ejphar.2007.01.059. Epub 2007 Feb 8. Eur J Pharmacol. 2007. PMID: 17349996 Free PMC article.
-
[Difference in tolerance to anti-hyperalgesic effect and its molecular mechanisms between chronic treatment with morphine, fentanyl and oxycodone in a chronic pain-like state].Nihon Shinkei Seishin Yakurigaku Zasshi. 2008 Nov;28(5-6):169-76. Nihon Shinkei Seishin Yakurigaku Zasshi. 2008. PMID: 19108502 Review. Japanese.
-
Ligand-Free Signaling of G-Protein-Coupled Receptors: Relevance to μ Opioid Receptors in Analgesia and Addiction.Molecules. 2022 Sep 8;27(18):5826. doi: 10.3390/molecules27185826. Molecules. 2022. PMID: 36144565 Free PMC article. Review.
Cited by
-
Allostatic Mechanisms of Opioid Tolerance Beyond Desensitization and Downregulation.Trends Pharmacol Sci. 2016 Nov;37(11):963-976. doi: 10.1016/j.tips.2016.08.002. Epub 2016 Sep 23. Trends Pharmacol Sci. 2016. PMID: 27670390 Free PMC article. Review.
-
Antinociceptive, reinforcing, and pruritic effects of the G-protein signalling-biased mu opioid receptor agonist PZM21 in non-human primates.Br J Anaesth. 2020 Oct;125(4):596-604. doi: 10.1016/j.bja.2020.06.057. Epub 2020 Aug 17. Br J Anaesth. 2020. PMID: 32819621 Free PMC article.
-
Fentanyl: Receptor pharmacology, abuse potential, and implications for treatment.Neurosci Biobehav Rev. 2019 Nov;106:49-57. doi: 10.1016/j.neubiorev.2018.12.005. Epub 2018 Dec 5. Neurosci Biobehav Rev. 2019. PMID: 30528374 Free PMC article. Review.
-
Treating Chronic Pain: An Overview of Clinical Studies Centered on the Buprenorphine Option.Drugs. 2018 Aug;78(12):1211-1228. doi: 10.1007/s40265-018-0953-z. Drugs. 2018. PMID: 30051169 Free PMC article. Review.
-
Opioid-Induced Pronociceptive Signaling in the Gastrointestinal Tract Is Mediated by Delta-Opioid Receptor Signaling.J Neurosci. 2022 Apr 20;42(16):3316-3328. doi: 10.1523/JNEUROSCI.2098-21.2022. Epub 2022 Mar 7. J Neurosci. 2022. PMID: 35256532 Free PMC article.
References
-
- Aceto MD, Bowman ER, Harris LS, May EL. Dependence Studies of New Compounds in the Rhesus Monkey, Rat and Mouse. In: Harris LS, editor. Problems of Drug Dependence, NIDA Research Monograph. Vol. 19997. NIDA; Washington D.C.: 1997. pp. 338–395.
-
- Arden JR, Segredo V, Wang Z, Lameh J, Sadee W. Phosphorylation and agonist-specific intracellular trafficking of an epitope-tagged mu-opioid receptor expressed in HEK 293 cells. J Neurochem. 1995;65:1636–1645. - PubMed
-
- Bailey CP, Smith FL, Kelly E, Dewey WL, Henderson G. How important is protein kinase C in mu-opioid receptor desensitization and morphine tolerance? Trends Pharmacol Sci. 2006;27:558–565. - PubMed
-
- Berge O-G, Garcia-Cabrera I, Hole K. Response latencies in the tail-flick test depend on tail skin temperature. Neurosci Lett. 1988;86:284–288. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

