Hypoxia-induced changes in pulmonary and systemic vascular resistance: where is the O2 sensor?
- PMID: 20713189
- PMCID: PMC2991475
- DOI: 10.1016/j.resp.2010.08.007
Hypoxia-induced changes in pulmonary and systemic vascular resistance: where is the O2 sensor?
Abstract
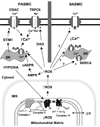
Pulmonary arteries (PA) constrict in response to alveolar hypoxia, whereas systemic arteries (SA) undergo dilation. These physiological responses reflect the need to improve gas exchange in the lung, and to enhance the delivery of blood to hypoxic systemic tissues. An important unresolved question relates to the underlying mechanism by which the vascular cells detect a decrease in oxygen tension and translate that into a signal that triggers the functional response. A growing body of work implicates the mitochondria, which appear to function as O2 sensors by initiating a redox-signaling pathway that leads to the activation of downstream effectors that regulate vascular tone. However, the direction of this redox signal has been the subject of controversy. Part of the problem has been the lack of appropriate tools to assess redox signaling in live cells. Recent advancements in the development of redox sensors have led to studies that help to clarify the nature of the hypoxia-induced redox signaling by reactive oxygen species (ROS). Moreover, these studies provide valuable insight regarding the basis for discrepancies in earlier studies of the hypoxia-induced mechanism of redox signaling. Based on recent work, it appears that the O2 sensing mechanism in both the PA and SA are identical, that mitochondria function as the site of O2 sensing, and that increased ROS release from these organelles leads to the activation of cell-specific, downstream vascular responses.
Copyright © 2010 Elsevier B.V. All rights reserved.
Figures


Similar articles
-
Diversity in mitochondrial function explains differences in vascular oxygen sensing.Circ Res. 2002 Jun 28;90(12):1307-15. doi: 10.1161/01.res.0000024689.07590.c2. Circ Res. 2002. PMID: 12089069
-
Increases in mitochondrial reactive oxygen species trigger hypoxia-induced calcium responses in pulmonary artery smooth muscle cells.Circ Res. 2006 Oct 27;99(9):970-8. doi: 10.1161/01.RES.0000247068.75808.3f. Epub 2006 Sep 28. Circ Res. 2006. PMID: 17008601
-
Molecular identification of O2 sensors and O2-sensitive potassium channels in the pulmonary circulation.Adv Exp Med Biol. 2000;475:219-40. doi: 10.1007/0-306-46825-5_21. Adv Exp Med Biol. 2000. PMID: 10849663 Review.
-
Ndufs2, a Core Subunit of Mitochondrial Complex I, Is Essential for Acute Oxygen-Sensing and Hypoxic Pulmonary Vasoconstriction.Circ Res. 2019 Jun 7;124(12):1727-1746. doi: 10.1161/CIRCRESAHA.118.314284. Epub 2019 Mar 29. Circ Res. 2019. PMID: 30922174 Free PMC article.
-
Hypoxic Pulmonary Vasoconstriction: An Important Component of the Homeostatic Oxygen Sensing System.Physiol Res. 2024 Nov 29;73(S2):S493-S510. doi: 10.33549/physiolres.935431. Physiol Res. 2024. PMID: 39589299 Free PMC article. Review.
Cited by
-
Long Range Endocrine Delivery of Circulating miR-210 to Endothelium Promotes Pulmonary Hypertension.Circ Res. 2020 Aug 14;127(5):677-692. doi: 10.1161/CIRCRESAHA.119.316398. Epub 2020 Jun 4. Circ Res. 2020. PMID: 32493166 Free PMC article.
-
Redox Signaling and Regional Heterogeneity of Endothelial Dysfunction in db/db Mice.Int J Mol Sci. 2020 Aug 26;21(17):6147. doi: 10.3390/ijms21176147. Int J Mol Sci. 2020. PMID: 32858910 Free PMC article.
-
Mitochondrial Regulation of the Hypoxia-Inducible Factor in the Development of Pulmonary Hypertension.J Clin Med. 2022 Sep 3;11(17):5219. doi: 10.3390/jcm11175219. J Clin Med. 2022. PMID: 36079149 Free PMC article. Review.
-
Dysregulation of the Nitric Oxide/Dimethylarginine Pathway in Hypoxic Pulmonary Vasoconstriction-Molecular Mechanisms and Clinical Significance.Front Med (Lausanne). 2022 Feb 17;9:835481. doi: 10.3389/fmed.2022.835481. eCollection 2022. Front Med (Lausanne). 2022. PMID: 35252268 Free PMC article. Review.
-
Exploring lung physiology in health and disease with lung slices.Pulm Pharmacol Ther. 2011 Oct;24(5):452-65. doi: 10.1016/j.pupt.2011.05.001. Epub 2011 May 12. Pulm Pharmacol Ther. 2011. PMID: 21600999 Free PMC article. Review.
References
-
- Aaronson PI, Robertson TP, Ward JP. Endothelium-derived mediators and hypoxic pulmonary vasoconstriction. Respir Physiolo Neurobiol. 2002;132:107–120. - PubMed
-
- Ali MH, Pearlstein DP, Mathieu CE, Schumacker PT. Mitochondrial Requirement for Endothelial Responses to Cyclic Strain: Implications for Mechanotransduction. Am J Physiol Lung Cell Mol Physiol. 2004 - PubMed
-
- Archer S, Michelakis E. The Mechanism(s) of Hypoxic Pulmonary Vasoconstriction: Potassium Channels, Redox O(2) Sensors, and Controversies. News Physiol Sci. 2002;17:131–137. - PubMed
-
- Archer SL, Huang J, Henry T, Peterson D, Weir EK. A redox-based O2 sensor in rat pulmonary vasculature. Circ Res. 1993;73:1100–1112. - PubMed
-
- Archer SL, London B, Hampl V, Wu X, Nsair A, Puttagunta L, Hashimoto K, Waite RE, Michelakis ED. Impairment of hypoxic pulmonary vasoconstriction in mice lacking the voltage-gated potassium channel Kv1.5. Faseb J. 2001;15:1801–1803. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

