Invasion of bovine peripheral blood mononuclear cells and erythrocytes by Mycoplasma bovis
- PMID: 20713619
- PMCID: PMC2976333
- DOI: 10.1128/IAI.00707-10
Invasion of bovine peripheral blood mononuclear cells and erythrocytes by Mycoplasma bovis
Abstract
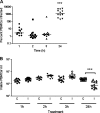
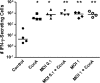
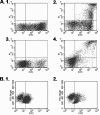
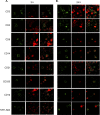
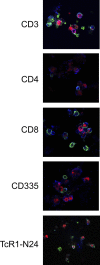
Mycoplasma bovis is a small, cell wall-less bacterium that contributes to a number of chronic inflammatory diseases in both dairy and feedlot cattle, including mastitis and bronchopneumonia. Numerous reports have implicated M. bovis in the activation of the immune system, while at the same time inhibiting immune cell proliferation. However, it is unknown whether the specific immune-cell population M. bovis is capable of attaching to and potentially invading. Here, we demonstrate that incubation of M. bovis Mb1 with bovine peripheral blood mononuclear cells (PBMC) resulted in a significant reduction in their proliferative responses while still remaining viable and capable of gamma interferon secretion. Furthermore, we show that M. bovis Mb1 can be found intracellularly (suggesting a role for either phagocytosis or attachment/invasion) in a number of select bovine PBMC populations (T cells, B cells, monocytes, γδ T cells, dendritic cells, NK cells, cytotoxic T cells, and T-helper cells), as well as red blood cells, albeit it at a significantly lower proportion. M. bovis Mb1 appeared to display three main patterns of intracellular staining: diffuse staining, an association with the intracellular side of the cell membrane, and punctate/vacuole-like staining. The invasion of circulating immune cells and erythrocytes could play an important role in disease pathogenesis by aiding the transport of M. bovis from the lungs to other sites.
Figures


Similar articles
-
Cytokine mRNA profiling and the proliferative response of bovine peripheral blood mononuclear cells to Mycoplasma bovis.Vet Immunol Immunopathol. 2015 May 15;165(1-2):45-53. doi: 10.1016/j.vetimm.2015.03.002. Epub 2015 Mar 6. Vet Immunol Immunopathol. 2015. PMID: 25800221
-
Mycoplasma bovis-Induced Inhibition of Bovine Peripheral Blood Mononuclear Cell Proliferation Is Ameliorated after Blocking the Immune-Inhibitory Programmed Death 1 Receptor.Infect Immun. 2018 Feb 20;86(3):e00921-17. doi: 10.1128/IAI.00921-17. Print 2018 Mar. Infect Immun. 2018. PMID: 29311234 Free PMC article.
-
Mycoplasma bovis isolates recovered from cattle and bison (Bison bison) show differential in vitro effects on PBMC proliferation, alveolar macrophage apoptosis and invasion of epithelial and immune cells.Vet Microbiol. 2016 Apr 15;186:28-36. doi: 10.1016/j.vetmic.2016.02.016. Epub 2016 Feb 23. Vet Microbiol. 2016. PMID: 27016754
-
Mycoplasma bovis pneumonia in cattle.Anim Health Res Rev. 2007 Dec;8(2):161-86. doi: 10.1017/S1466252307001351. Anim Health Res Rev. 2007. PMID: 18218159 Review.
-
Pathogenesis and Virulence of Mycoplasma bovis.Vet Clin North Am Food Anim Pract. 2020 Jul;36(2):269-278. doi: 10.1016/j.cvfa.2020.02.002. Epub 2020 Apr 21. Vet Clin North Am Food Anim Pract. 2020. PMID: 32327249 Review.
Cited by
-
Novel Secreted Protein of Mycoplasma bovis MbovP280 Induces Macrophage Apoptosis Through CRYAB.Front Immunol. 2021 Feb 15;12:619362. doi: 10.3389/fimmu.2021.619362. eCollection 2021. Front Immunol. 2021. PMID: 33659004 Free PMC article.
-
Mycoplasmas as Host Pantropic and Specific Pathogens: Clinical Implications, Gene Transfer, Virulence Factors, and Future Perspectives.Front Cell Infect Microbiol. 2022 May 13;12:855731. doi: 10.3389/fcimb.2022.855731. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35646746 Free PMC article. Review.
-
Bovine Epithelial in vitro Infection Models for Mycoplasma bovis.Front Cell Infect Microbiol. 2018 Sep 18;8:329. doi: 10.3389/fcimb.2018.00329. eCollection 2018. Front Cell Infect Microbiol. 2018. PMID: 30280094 Free PMC article.
-
Mycoplasma pneumoniae downregulates RECK to promote matrix metalloproteinase-9 secretion by bronchial epithelial cells.Virulence. 2022 Dec;13(1):1270-1284. doi: 10.1080/21505594.2022.2101746. Virulence. 2022. PMID: 35892136 Free PMC article.
-
The Putative Role of Viruses, Bacteria, and Chronic Fungal Biotoxin Exposure in the Genesis of Intractable Fatigue Accompanied by Cognitive and Physical Disability.Mol Neurobiol. 2016 May;53(4):2550-71. doi: 10.1007/s12035-015-9262-7. Epub 2015 Jun 17. Mol Neurobiol. 2016. PMID: 26081141 Review.
References
-
- Baseman, J. B., S. P. Reddy, and S. F. Dallo. 1996. Interplay between mycoplasma surface proteins, airway cells, and the protean manifestations of mycoplasma-mediated human infections. Am. J. Respir. Crit. Care Med. 154:S137-S144. - PubMed
-
- Bendelac, A., P. B. Savage, and L. Teyton. 2007. The Biology of NKT Cells. Annu. Rev. Immunol. 25:297-336. - PubMed
-
- Booker, C. W., S. M. Abutarbush, P. S. Morley, G. K. Jim, T. J. Pittman, O. C. Schunicht, T. Perrett, B. K. Wildman, R. K. Fenton, P. T. Guichon, and E. D. Janzen. 2008. Microbiological and histopathological findings in cases of fatal bovine respiratory disease of feedlot cattle in Western Canada. Can. Vet. J. 49:473-481. - PMC - PubMed
-
- Boothby, J. T., D. E. Jasper, J. G. Zinkl, C. B. Thomas, and J. D. Dellinger. 1983. Prevalence of mycoplasmas and immune responses to Mycoplasma bovis in feedlot calves. Am. J. Vet. Res. 44:831-838. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

