Chalcones and dihydrochalcones augment TRAIL-mediated apoptosis in prostate cancer cells
- PMID: 20714300
- PMCID: PMC6257745
- DOI: 10.3390/molecules15085336
Chalcones and dihydrochalcones augment TRAIL-mediated apoptosis in prostate cancer cells
Abstract
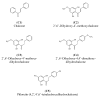
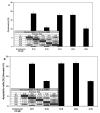
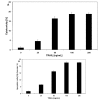
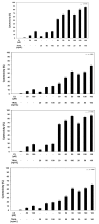
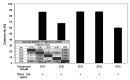
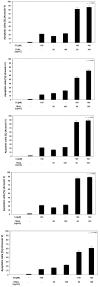
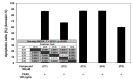
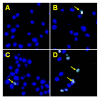
Chalcones and dihydrochalcones exhibit chemopreventive and antitumor activity. TRAIL (tumor necrosis factor-related apoptosis-inducing ligand) is a natural endogenous anticancer agent. We examined the cytotoxic and apoptotic effect of chalcones and dihydrochalcones on TRAIL-mediated apoptosis in LNCaP prostate cancer cells. The cytotoxicity was evaluated by the MTT and LDH assays. The apoptosis was detected using annexin V-FITC by flow cytometry and fluorescence microscopy. The DeltaPsim was evaluated using DePsipher staining by fluorescence microscopy. Our study showed that two tested chalcones (chalcone and 2',6'dihydroxy-4'-methoxychalcone) and three dihydrochalcones (2',6'-dihydroxy-4'4-dimethoxydihydrochalcone, 2',6'-dihydroxy-4'-methoxydihydro- chalcone, and 2',4',6'-trihydroxydihydrochalcone, called phloretin) markedly augmented TRAIL-induced apoptosis and cytotoxicity in LNCaP cells and confirmed the significant role of chalcones in chemoprevention of prostate cancer.
Figures
Similar articles
-
Chalcones enhance TRAIL-induced apoptosis in prostate cancer cells.Int J Mol Sci. 2009 Dec 24;11(1):1-13. doi: 10.3390/ijms11010001. Int J Mol Sci. 2009. PMID: 20161998 Free PMC article.
-
The dietary flavonol fisetin enhances the apoptosis-inducing potential of TRAIL in prostate cancer cells.Int J Oncol. 2011 Oct;39(4):771-9. doi: 10.3892/ijo.2011.1116. Epub 2011 Jul 6. Int J Oncol. 2011. PMID: 21743964
-
Ethanolic extract of Brazilian green propolis sensitizes prostate cancer cells to TRAIL-induced apoptosis.Int J Oncol. 2011 Apr;38(4):941-53. doi: 10.3892/ijo.2011.930. Epub 2011 Feb 1. Int J Oncol. 2011. PMID: 21286663
-
Molecular mechanisms underlying chemopreventive potential of butein: Current trends and future perspectives.Chem Biol Interact. 2021 Dec 1;350:109699. doi: 10.1016/j.cbi.2021.109699. Epub 2021 Oct 11. Chem Biol Interact. 2021. PMID: 34648814 Review.
-
Dihydrochalcones as Antitumor Agents.Curr Med Chem. 2022 Aug 15;29(30):5042-5061. doi: 10.2174/0929867329666220415113219. Curr Med Chem. 2022. PMID: 35430969 Review.
Cited by
-
Prima-1 induces apoptosis in bladder cancer cell lines by activating p53.Clinics (Sao Paulo). 2013;68(3):297-303. doi: 10.6061/clinics/2013(03)oa03. Clinics (Sao Paulo). 2013. PMID: 23644847 Free PMC article.
-
Antimicrobial, Antioxidant, and Anticancer Activities of Some Novel Isoxazole Ring Containing Chalcone and Dihydropyrazole Derivatives.Molecules. 2020 Feb 26;25(5):1047. doi: 10.3390/molecules25051047. Molecules. 2020. PMID: 32110945 Free PMC article.
-
NutriTRAILomics in prostate cancer: time to have two strings to one's bow.Mol Biol Rep. 2012 Apr;39(4):4909-14. doi: 10.1007/s11033-011-1286-0. Epub 2011 Dec 6. Mol Biol Rep. 2012. PMID: 22143879 Review.
-
Chlorin-based photodynamic therapy enhances the effect of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) in bladder cancer cells.Med Sci Monit. 2012 Jan;18(1):BR47-53. doi: 10.12659/msm.882203. Med Sci Monit. 2012. PMID: 22207109 Free PMC article.
-
Synthesis, characterization, and biological activity of a fresh class of sonochemically synthesized Cu2+ complexes.Sci Rep. 2024 Sep 12;14(1):21325. doi: 10.1038/s41598-024-72345-8. Sci Rep. 2024. PMID: 39266594 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

