Efficient generation of functional dopaminergic neurons from human induced pluripotent stem cells under defined conditions
- PMID: 20715183
- PMCID: PMC2996088
- DOI: 10.1002/stem.499
Efficient generation of functional dopaminergic neurons from human induced pluripotent stem cells under defined conditions
Abstract
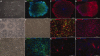
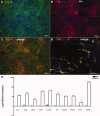
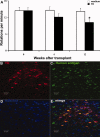
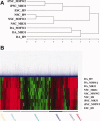
Human induced pluripotent stem cells (iPSCs) reprogrammed from somatic cells represent a promising unlimited cell source for generating patient-specific cells for biomedical research and personalized medicine. As a first step, critical to clinical applications, we attempted to develop defined culture conditions to expand and differentiate human iPSCs into functional progeny such as dopaminergic neurons for treating or modeling Parkinson's disease (PD). We used a completely defined (xeno-free) system that we previously developed for efficient generation of authentic dopaminergic neurons from human embryonic stem cells (hESCs), and applied it to iPSCs. First, we adapted two human iPSC lines derived from different somatic cell types for the defined expansion medium and showed that the iPSCs grew similarly as hESCs in the same medium regarding pluripotency and genomic stability. Second, by using these two independent adapted iPSC lines, we showed that the process of differentiation into committed neural stem cells (NSCs) and subsequently into dopaminergic neurons was also similar to hESCs. Importantly, iPSC-derived dopaminergic neurons were functional as they survived and improved behavioral deficits in 6-hydroxydopamine-leasioned rats after transplantation. In addition, iPSC-derived NSCs and neurons could be efficiently transduced by a baculoviral vector delivering episomal DNA for future gene function study and disease modeling using iPSCs. We also performed genome-wide microarray comparisons between iPSCs and hESCs, and we derived NSC and dopaminergic neurons. Our data revealed overall similarity and visible differences at a molecular level. Efficient generation of functional dopaminergic neurons under defined conditions will facilitate research and applications using PD patient-specific iPSCs.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

