Characterization of the molecular mechanism underlying gibberellin perception complex formation in rice
- PMID: 20716699
- PMCID: PMC2947161
- DOI: 10.1105/tpc.110.075549
Characterization of the molecular mechanism underlying gibberellin perception complex formation in rice
Abstract
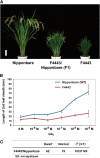
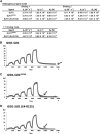
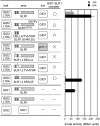
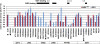
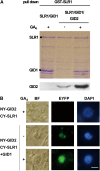
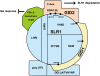
The DELLA protein SLENDER RICE1 (SLR1) is a repressor of gibberellin (GA) signaling in rice (Oryza sativa), and most of the GA-associated responses are induced upon SLR1 degradation. It is assumed that interaction between GIBBERELLIN INSENSITIVE DWARF1 (GID1) and the N-terminal DELLA/TVHYNP motif of SLR1 triggers F-box protein GID2-mediated SLR1 degradation. We identified a semidominant dwarf mutant, Slr1-d4, which contains a mutation in the region encoding the C-terminal GRAS domain of SLR1 (SLR1(G576V)). The GA-dependent degradation of SLR1(G576V) was reduced in Slr1-d4, and compared with SLR1, SLR1(G576V) showed reduced interaction with GID1 and almost none with GID2 when tested in yeast cells. Surface plasmon resonance of GID1-SLR1 and GID1-SLR1(G576V) interactions revealed that the GRAS domain of SLR1 functions to stabilize the GID1-SLR1 interaction by reducing its dissociation rate and that the G576V substitution in SLR1 diminishes this stability. These results suggest that the stable interaction of GID1-SLR1 through the GRAS domain is essential for the recognition of SLR1 by GID2. We propose that when the DELLA/TVHYNP motif of SLR1 binds with GID1, it enables the GRAS domain of SLR1 to interact with GID1 and that the stable GID1-SLR1 complex is efficiently recognized by GID2.
Figures




References
-
- Abe M., Kobayashi Y., Yamamoto S., Daimon Y., Yamaguchi A., Ikeda Y., Ichinoki H., Notaguchi M., Goto K., Araki T. (2005). FD, a bZIP protein mediating signals from the floral pathway integrator FT at the shoot apex. Science 309: 1052–1056 - PubMed
-
- Aleman L., Kitamura J., Abdel-mageed H., Lee J., Sun Y., Nakajima M., Ueguchi-Tanaka M., Matsuoka M., Allen R.D. (2008). Functional analysis of cotton orthologs of GA signal transduction factors GID1 and SLR1. Plant Mol. Biol. 68: 1–16 - PubMed
-
- Asano K., Hirano K., Ueguchi-Tanaka M., Angeles-Shim R.B., Komura T., Satoh H., Kitano H., Matsuoka M., Ashikari M. (2009). Isolation and characterization of dominant dwarf mutants, Slr1-d, in rice. Mol. Genet. Genomics 281: 223–231 - PubMed
-
- Bassel G.W., Mullen R.T., Bewley J.D. (2008). procera is a putative DELLA mutant in tomato (Solanum lycopersicum): Effects on the seed and vegetative plant. J. Exp. Bot. 59: 585–593 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

