Development of a novel mammalian cell surface antibody display platform
- PMID: 20716968
- PMCID: PMC2958572
- DOI: 10.4161/mabs.2.5.12970
Development of a novel mammalian cell surface antibody display platform
Abstract
Antibody display systems have been successfully applied to screen, select and characterize antibody fragments. These systems typically use prokaryotic organisms such as phage and bacteria or lower eukaryotic organisms, such as yeast. These organisms possess either no or different post-translational modification functions from mammalian cells and prefer to display small antibody fragments instead of full-length IgGs. We report here a novel mammalian cell-based antibody display platform that displays full-length functional antibodies on the surface of mammalian cells. Through recombinase-mediated DNA integration, each host cell contains one copy of the gene of interest in the genome. Utilizing a hot-spot integration site, the expression levels of the gene of interest are high and comparable between clones, ensuring a high signal to noise ratio. Coupled with fluorescence-activated cell sorting (FACS) technology, our platform is high throughput and can distinguish antibodies with very high antigen binding affinities directly on the cell surface. Single-round FACS can enrich high affinity antibodies by more than 500 fold. Antibodies with significantly improved neutralizing activity have been identified from a randomly mutagenized library, demonstrating the power of this platform in screening and selecting antibody therapeutics.
Figures



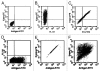

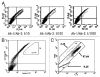


References
-
- Aires DSF, Corte-Real S, Goncalves J. Recombinant antibodies as therapeutic agents: pathways for modeling new biodrugs. BioDrugs. 2008;22:301–314. - PubMed
-
- Reichert JM, Rosensweig CJ, Faden LB, Dewitz MC. Monoclonal antibody successes in the clinic. Nat Biotechnol. 2005;23:1073–1078. - PubMed
-
- Smith GP. Filamentous fusion phage: novel expression vectors that display cloned antigens on the virion surface. Science. 1985;228:1315–1317. - PubMed
-
- Winter G, Griffiths AD, Hawkins RE, Hoogenboom HR. Making antibodies by phage display. Annu Rev Immunol. 1994;12:433–455. - PubMed
-
- Low NM, Holliger P, Winter G. Mimicking somatic hypermutation: Affinity maturation of antibodies displayed on bacteriophage using a bacterial mutator strain. J Mol Biol. 1996;260:359–368. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
