Spatial organization of adhesion: force-dependent regulation and function in tissue morphogenesis
- PMID: 20717145
- PMCID: PMC2924654
- DOI: 10.1038/emboj.2010.182
Spatial organization of adhesion: force-dependent regulation and function in tissue morphogenesis
Abstract
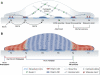
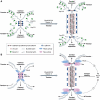
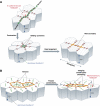
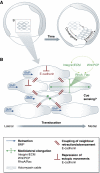
Integrin- and cadherin-mediated adhesion is central for cell and tissue morphogenesis, allowing cells and tissues to change shape without loosing integrity. Studies predominantly in cell culture showed that mechanosensation through adhesion structures is achieved by force-mediated modulation of their molecular composition. The specific molecular composition of adhesion sites in turn determines their signalling activity and dynamic reorganization. Here, we will review how adhesion sites respond to mecanical stimuli, and how spatially and temporally regulated signalling from different adhesion sites controls cell migration and tissue morphogenesis.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

References
-
- Anastasiadis PZ, Moon SY, Thoreson MA, Mariner DJ, Crawford HC, Zheng Y, Reynolds AB (2000) Inhibition of RhoA by p120 catenin. Nat Cell Biol 2: 637–644 - PubMed
-
- Arboleda-Estudillo Y, Krieg M, Stuhmer J, Licata NA, Muller DJ, Heisenberg CP (2010) Movement directionality in collective migration of germ layer progenitors. Curr Biol 20: 161–169 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

