Robust anti-arrhythmic efficacy of verapamil and flunarizine against dofetilide-induced TdP arrhythmias is based upon a shared and a different mode of action
- PMID: 20718748
- PMCID: PMC2962825
- DOI: 10.1111/j.1476-5381.2010.00883.x
Robust anti-arrhythmic efficacy of verapamil and flunarizine against dofetilide-induced TdP arrhythmias is based upon a shared and a different mode of action
Abstract
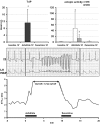
Background and purpose: The high predisposition to Torsade de Pointes (TdP) in dogs with chronic AV-block (CAVB) is well documented. The anti-arrhythmic efficacy and mode of action of Ca(2+) channel antagonists, flunarizine and verapamil against TdP were investigated.
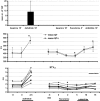
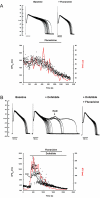
Experimental approach: Mongrel dogs with CAVB were selected based on the inducibility of TdP with dofetilide. The effects of flunarizine and verapamil were assessed after TdP and in different experiments to prevent dofetilide-induced TdP. Electrocardiogram and ventricular monophasic action potentials were recorded. Electrophysiological parameters and short-term variability of repolarization (STV) were determined. In vitro, flunarizine and verapamil were added to determine their effect on (i) dofetilide-induced early after depolarizations (EADs) in canine ventricular myocytes (VM); (ii) diastolic Ca(2+) sparks in RyR2(R4496+/+) mouse myocytes; and (iii) peak and late I(Na) in SCN5A-HEK 293 cells.
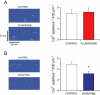
Key results: Dofetilide increased STV prior to TdP and in VM prior to EADs. Both flunarizine and verapamil completely suppressed TdP and reversed STV to baseline values. Complete prevention of TdP was achieved with both drugs, accompanied by the prevention of an increase in STV. Suppression of EADs was confirmed after flunarizine. Only flunarizine blocked late I(Na). Ca(2+) sparks were reduced with verapamil.
Conclusions and implications: Robust anti-arrhythmic efficacy was seen with both Ca(2+) channel antagonists. Their divergent electrophysiological actions may be related to different additional effects of the two drugs.
Figures
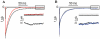
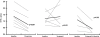
Similar articles
-
Short-term Variability of Repolarization Is Superior to Other Repolarization Parameters in the Evaluation of Diverse Antiarrhythmic Interventions in the Chronic Atrioventricular Block Dog.J Cardiovasc Pharmacol. 2017 Jun;69(6):398-407. doi: 10.1097/FJC.0000000000000488. J Cardiovasc Pharmacol. 2017. PMID: 28574954 Free PMC article. Review.
-
Istaroxime, a positive inotropic agent devoid of proarrhythmic properties in sensitive chronic atrioventricular block dogs.Pharmacol Res. 2018 Jul;133:132-140. doi: 10.1016/j.phrs.2018.05.001. Epub 2018 May 16. Pharmacol Res. 2018. PMID: 29753687
-
Selective late sodium current inhibitor GS-458967 suppresses Torsades de Pointes by mostly affecting perpetuation but not initiation of the arrhythmia.Br J Pharmacol. 2018 Jun;175(12):2470-2482. doi: 10.1111/bph.14217. Epub 2018 May 6. Br J Pharmacol. 2018. PMID: 29582428 Free PMC article.
-
Point of View: Electrophysiological Endpoints Differ When Comparing the Mode of Action of Highly Successful Anti-arrhythmic Drugs in the CAVB Dog Model With TdP.J Cardiovasc Pharmacol. 2019 Dec;74(6):499-507. doi: 10.1097/FJC.0000000000000748. J Cardiovasc Pharmacol. 2019. PMID: 31738198 Review.
-
Late na(+) current inhibition by ranolazine reduces torsades de pointes in the chronic atrioventricular block dog model.J Am Coll Cardiol. 2010 Feb 23;55(8):801-9. doi: 10.1016/j.jacc.2009.10.033. J Am Coll Cardiol. 2010. PMID: 20170820
Cited by
-
Short-term Variability of Repolarization Is Superior to Other Repolarization Parameters in the Evaluation of Diverse Antiarrhythmic Interventions in the Chronic Atrioventricular Block Dog.J Cardiovasc Pharmacol. 2017 Jun;69(6):398-407. doi: 10.1097/FJC.0000000000000488. J Cardiovasc Pharmacol. 2017. PMID: 28574954 Free PMC article. Review.
-
The canine chronic atrioventricular block model in cardiovascular preclinical drug research.Br J Pharmacol. 2022 Mar;179(5):859-881. doi: 10.1111/bph.15436. Epub 2021 May 4. Br J Pharmacol. 2022. PMID: 33684961 Free PMC article. Review.
-
Arrhythmia Inducibility in the CAVB Dog Model, A Critical Analysis on Underlying Factors.Cardiovasc Toxicol. 2025 Sep;25(9):1344-1351. doi: 10.1007/s12012-025-10033-3. Epub 2025 Jun 24. Cardiovasc Toxicol. 2025. PMID: 40553351 Free PMC article.
-
The electromechanical window is no better than QT prolongation to assess risk of Torsade de Pointes in the complete atrioventricular block model in dogs.Br J Pharmacol. 2014 Feb;171(3):714-22. doi: 10.1111/bph.12483. Br J Pharmacol. 2014. PMID: 24490860 Free PMC article.
-
Verapamil as an antiarrhythmic agent in congestive heart failure: hopping from rabbit to human?Br J Pharmacol. 2012 May;166(2):554-6. doi: 10.1111/j.1476-5381.2011.01818.x. Br J Pharmacol. 2012. PMID: 22188337 Free PMC article.
References
-
- Aiba T, Shimizu W, Inagaki M, Noda T, Miyoshi S, Ding WG, et al. Cellular and ionic mechanism for drug-induced long QT syndrome and effectiveness of verapamil. J Am Coll Cardiol. 2005;45:300–307. - PubMed
-
- Anderson ME, Braun AP, Wu Y, Lu T, Wu Y, Schulman H, et al. KN-93, an inhibitor of multifunctional Ca++/calmodulin-dependent protein kinase, decreases early afterdepolarizations in rabbit heart. J Pharmacol Exp Ther. 1998;287:996–1006. - PubMed
-
- Antoons G, Oros A, Beekman JDM, Engelen MA, Houtman MJ, Belardinelli L, et al. Late Na+ current inhibition by Ranolazine reduces Torsades de Pointes in the chronic atrioventricular block dog model. J Am Coll Cardiol. 2010;55:801–809. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

