Regulation of interleukin-6 expression in human decidual cells and its potential role in chorioamnionitis
- PMID: 20724602
- PMCID: PMC2947272
- DOI: 10.2353/ajpath.2010.090781
Regulation of interleukin-6 expression in human decidual cells and its potential role in chorioamnionitis
Abstract
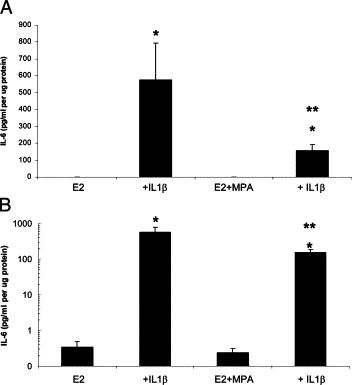
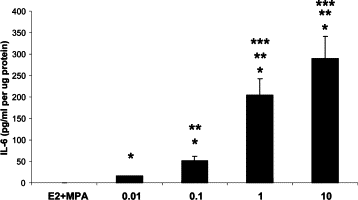
Chorioamnionitis frequently precedes both genital tract and placental inflammation and is both a primary cause of maternal morbidity and a major antecedent of preterm premature rupture of the membranes (PPROM) as well as preterm delivery (PTD). In most cases of chorioamnionitis, neutrophils dominate the decidua. In a subset of these cases, a predominance of monocytes is uniquely associated with both neonatal intraventricular hemorrhage and death. The multifunctional cytokine, interleukin-6, promotes local monocyte dominance via several mechanisms. In this study, immunostaining of placental sections revealed significantly higher interleukin-6 HSCOREs in decidual cells (DCs) but not in interstitial trophoblasts, in chorioamnionitis versus gestational age-matched control placentas (P < 0.05). In confluent leukocyte-free term DCs, secreted interleukin-6 levels in incubations with estradiol-17β were increased 2500-fold by IL-1β (P < 0.05). This up-regulation was inhibited by more than 50% in parallel incubations that included medroxyprogesterone acetate (n = 12, P < 0.05). Western blotting data confirmed these enzyme-linked immunosorbent assay results; quantitative RT-PCR findings demonstrated corresponding changes in interleukin-6 mRNA levels. Specific inhibitors of signaling for both nuclear factor-κB activation and p38-mitogen-activated protein kinase, but not for protein kinase C, significantly decreased IL-1β-enhanced interleukin-6 expression levels in cultured DCs. In conclusion, in situ and in vitro results indicate that significantly enhanced interleukin-6 expression levels in DCs during chorioamnionitis could be pivotal in skewing decidual monocyte differentiation to macrophages.
Figures



Similar articles
-
Progestin-inflammatory cytokine interactions affect matrix metalloproteinase-1 and -3 expression in term decidual cells: implications for treatment of chorioamnionitis-induced preterm delivery.J Clin Endocrinol Metab. 2008 Jan;93(1):252-9. doi: 10.1210/jc.2007-1538. Epub 2007 Oct 16. J Clin Endocrinol Metab. 2008. PMID: 17940116 Free PMC article.
-
Tumor necrosis factor-alpha and interleukin-1beta regulate interleukin-8 expression in third trimester decidual cells: implications for the genesis of chorioamnionitis.Am J Pathol. 2006 Oct;169(4):1294-302. doi: 10.2353/ajpath.2006.060185. Am J Pathol. 2006. PMID: 17003486 Free PMC article.
-
Mechanisms of abruption-induced premature rupture of the fetal membranes: thrombin-enhanced interleukin-8 expression in term decidua.Am J Pathol. 2005 Nov;167(5):1443-9. doi: 10.1016/S0002-9440(10)61230-8. Am J Pathol. 2005. PMID: 16251427 Free PMC article.
-
Regulation of monocyte chemoattractant protein-1 expression by tumor necrosis factor-alpha and interleukin-1beta in first trimester human decidual cells: implications for preeclampsia.Am J Pathol. 2006 Feb;168(2):445-52. doi: 10.2353/ajpath.2006.050082. Am J Pathol. 2006. PMID: 16436659 Free PMC article.
-
Acute chorioamnionitis and funisitis: definition, pathologic features, and clinical significance.Am J Obstet Gynecol. 2015 Oct;213(4 Suppl):S29-52. doi: 10.1016/j.ajog.2015.08.040. Am J Obstet Gynecol. 2015. PMID: 26428501 Free PMC article. Review.
Cited by
-
Interleukin (IL)-6: A Friend or Foe of Pregnancy and Parturition? Evidence From Functional Studies in Fetal Membrane Cells.Front Physiol. 2020 Jul 24;11:891. doi: 10.3389/fphys.2020.00891. eCollection 2020. Front Physiol. 2020. PMID: 32848846 Free PMC article.
-
Regulation of tryptophan 2,3-dioxygenase by HOXA10 enhances embryo viability through serotonin signaling.Am J Physiol Endocrinol Metab. 2011 Jan;300(1):E86-93. doi: 10.1152/ajpendo.00439.2010. Epub 2010 Oct 19. Am J Physiol Endocrinol Metab. 2011. PMID: 20959529 Free PMC article.
-
IL-1 and TNF mediates IL-6 signaling at the maternal-fetal interface during intrauterine inflammation.Front Immunol. 2024 Jun 4;15:1416162. doi: 10.3389/fimmu.2024.1416162. eCollection 2024. Front Immunol. 2024. PMID: 38895127 Free PMC article.
-
Translationally controlled tumour protein (TCTP) is present in human cornea and increases in herpetic keratitis.Diagn Pathol. 2012 Aug 1;7:90. doi: 10.1186/1746-1596-7-90. Diagn Pathol. 2012. PMID: 22853445 Free PMC article.
-
Neonatal posthemorrhagic hydrocephalus from prematurity: pathophysiology and current treatment concepts.J Neurosurg Pediatr. 2012 Mar;9(3):242-58. doi: 10.3171/2011.12.PEDS11136. J Neurosurg Pediatr. 2012. PMID: 22380952 Free PMC article. Review.
References
-
- Mueller-Heubach E, Rubinstein DN, Schwarz SS. Histologic chorioamnionitis and preterm delivery in different patient populations. Obstet Gynecol. 1990;75:622–626. - PubMed
-
- Hamilton BE, Martin JA, Sutton PD. Births: preliminary data for 2003. Natl Vital Stat Rep. 2004;53:1–17. - PubMed
-
- Newton ER. Preterm labor, preterm premature rupture of membranes, and chorioamnionitis. Clin Perinatol. 2005;32:571–600. - PubMed
-
- Romero R, Mazor M, Wu YK, Sirtori M, Oyarzun E, Mitchell MD, Hobbins JC. Infection in the pathogenesis of preterm labor. Semin Perinatol. 1988;12:262–279. - PubMed
-
- Shim SS, Romero R, Hong JS, Park CW, Jun JK, Kim BI, Yoon BH. Clinical significance of intra-amniotic inflammation in patients with preterm premature rupture of membranes. Am J Obstet Gynecol. 2004;191:1339–1345. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

