Aquaporin-3 mediates hydrogen peroxide uptake to regulate downstream intracellular signaling
- PMID: 20724658
- PMCID: PMC2936599
- DOI: 10.1073/pnas.1005776107
Aquaporin-3 mediates hydrogen peroxide uptake to regulate downstream intracellular signaling
Abstract
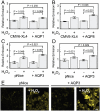
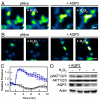
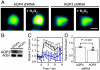
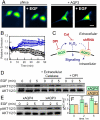
Hydrogen peroxide (H(2)O(2)) produced by cell-surface NADPH Oxidase (Nox) enzymes is emerging as an important signaling molecule for growth, differentiation, and migration processes. However, how cells spatially regulate H(2)O(2) to achieve physiological redox signaling over nonspecific oxidative stress pathways is insufficiently understood. Here we report that the water channel Aquaporin-3 (AQP3) can facilitate the uptake of H(2)O(2) into mammalian cells and mediate downstream intracellular signaling. Molecular imaging with Peroxy Yellow 1 Methyl-Ester (PY1-ME), a new chemoselective fluorescent indicator for H(2)O(2), directly demonstrates that aquaporin isoforms AQP3 and AQP8, but not AQP1, can promote uptake of H(2)O(2) specifically through membranes in mammalian cells. Moreover, we show that intracellular H(2)O(2) accumulation can be modulated up or down based on endogenous AQP3 expression, which in turn can influence downstream cell signaling cascades. Finally, we establish that AQP3 is required for Nox-derived H(2)O(2) signaling upon growth factor stimulation. Taken together, our findings demonstrate that the downstream intracellular effects of H(2)O(2) can be regulated across biological barriers, a discovery that has broad implications for the controlled use of this potentially toxic small molecule for beneficial physiological functions.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Poole LB, Karplus PA, Claiborne A. Protein sulfenic acids in redox signaling. Annu Rev Pharmacol Toxicol. 2004;44:325–347. - PubMed
-
- Rhee SG. Cell signaling H2O2, a necessary evil for cell signaling. Science. 2006;312:1882–1883. - PubMed
-
- D’Autreaux B, Toledano MB. ROS as signalling molecules: mechanisms that generate specificity in ROS homeostasis. Nat Rev Mol Cell Biol. 2007;8:813–824. - PubMed
-
- Winterbourn CC. Reconciling the chemistry and biology of reactive oxygen species. Nat Chem Biol. 2008;4:278–286. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

