Akt2 deficiency promotes cardiac induction of Rab4a and myocardial β-adrenergic hypersensitivity
- PMID: 20728450
- PMCID: PMC2975863
- DOI: 10.1016/j.yjmcc.2010.08.011
Akt2 deficiency promotes cardiac induction of Rab4a and myocardial β-adrenergic hypersensitivity
Abstract
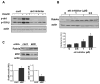
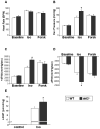
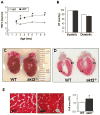
Patients with diabetes mellitus can develop cardiac dysfunction in the absence of underlying coronary artery disease or hypertension; a condition defined as diabetic cardiomyopathy. Mice lacking the intracellular protein kinase Akt2 develop a syndrome that is similar to diabetes mellitus type 2. Expression profiling of akt2(-/-) myocardium revealed that Rab4a, a GTPase involved in glucose transporter 4 translocation and β-adrenergic receptor (βAR) recycling to the plasma membrane, was significantly induced. We therefore hypothesized that Akt2 deficiency increases myocardial β-adrenergic sensitivity. Confirmatory analysis revealed up-regulation of Rab4a mRNA and protein in akt2(-/-) myocardium. In cultured cardiomyocyte experiments, Rab4a was induced by pharmacological inhibition of Akt as well as by specific knockdown of Akt2 with siRNA. Isolated akt2(-/-) hearts were hypersensitive to isoproterenol (ISO) but exhibited normal sensitivity to forskolin. Prolonged ISO treatment led to increased cardiac hypertrophy in akt2(-/-) mice compared to wild type mice. In addition, spontaneous hypertrophy was noted in aged akt2(-/-) hearts that was inhibited by treatment with the βAR blocker propranolol. In agreement with previous results demonstrating increased fatty acid oxidation rates in akt2(-/-) myocardium, we found increased peroxisome proliferator-activated receptor α (PPARα) activity in the hearts of these animals. Interestingly, increased myocardial Rab4a expression was present in mice with cardiac-specific overexpression of PPARα and was also observed upon stimulation of PPARα activity in cultured cardiomyocytes. Accordingly, propranolol attenuated the development of cardiac hypertrophy in the PPARα transgenic mice as well. Our results indicate that reduced Akt2 leads to up-regulation of Rab4a expression in cardiomyocytes in a cell-autonomous fashion that may involve activation of PPARα. This maladaptive response is associated with hypersensitivity of akt2(-/-) myocardium to β-adrenergic stimulation.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Figures





Similar articles
-
Rab4a signaling unmasks a pivotal link between myocardial homeostasis and metabolic remodeling in the diabetic heart.J Mol Cell Cardiol. 2010 Dec;49(6):908-10. doi: 10.1016/j.yjmcc.2010.09.002. Epub 2010 Sep 16. J Mol Cell Cardiol. 2010. PMID: 20840849 Free PMC article. No abstract available.
-
Enhancement of the recycling and activation of beta-adrenergic receptor by Rab4 GTPase in cardiac myocytes.J Biol Chem. 2006 Apr 21;281(16):11097-103. doi: 10.1074/jbc.M511460200. Epub 2006 Feb 16. J Biol Chem. 2006. PMID: 16484224 Free PMC article.
-
Interplay between the E2F pathway and β-adrenergic signaling in the pathological hypertrophic response of myocardium.J Mol Cell Cardiol. 2015 Jul;84:179-90. doi: 10.1016/j.yjmcc.2015.04.026. Epub 2015 May 2. J Mol Cell Cardiol. 2015. PMID: 25944088
-
Metallothionein Preserves Akt2 Activity and Cardiac Function via Inhibiting TRB3 in Diabetic Hearts.Diabetes. 2018 Mar;67(3):507-517. doi: 10.2337/db17-0219. Epub 2017 Oct 27. Diabetes. 2018. PMID: 29079702 Free PMC article.
-
Regulation of phospholamban and troponin-I phosphorylation in the intact rat cardiomyocytes by adrenergic and cholinergic stimuli: roles of cyclic nucleotides, calcium, protein kinases and phosphatases and depolarization.Mol Cell Biochem. 1995 Aug-Sep;149-150:103-26. doi: 10.1007/BF01076569. Mol Cell Biochem. 1995. PMID: 8569720 Review.
Cited by
-
Exercise training and PI3Kα-induced electrical remodeling is independent of cellular hypertrophy and Akt signaling.J Mol Cell Cardiol. 2012 Oct;53(4):532-41. doi: 10.1016/j.yjmcc.2012.07.004. Epub 2012 Jul 21. J Mol Cell Cardiol. 2012. PMID: 22824041 Free PMC article.
-
zDHHC9 Regulates Cardiomyocyte Rab3a Activity and Atrial Natriuretic Peptide Secretion Through Palmitoylation of Rab3gap1.JACC Basic Transl Sci. 2023 Feb 22;8(5):518-542. doi: 10.1016/j.jacbts.2022.11.003. eCollection 2023 May. JACC Basic Transl Sci. 2023. PMID: 37325411 Free PMC article.
-
Cardiovascular Dysfunction Following Burn Injury: What We Have Learned from Rat and Mouse Models.Int J Mol Sci. 2016 Jan 2;17(1):53. doi: 10.3390/ijms17010053. Int J Mol Sci. 2016. PMID: 26729111 Free PMC article. Review.
-
Peptidomic analysis of follicular fluid in patients with polycystic ovarian syndrome.Front Cell Dev Biol. 2023 Nov 8;11:1289063. doi: 10.3389/fcell.2023.1289063. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 38020909 Free PMC article.
-
Dock10 Regulates Cardiac Function under Neurohormonal Stress.Int J Mol Sci. 2022 Aug 25;23(17):9616. doi: 10.3390/ijms23179616. Int J Mol Sci. 2022. PMID: 36077014 Free PMC article.
References
-
- Cohen-Solal A, Beauvais F, Logeart D. Heart failure and diabetes mellitus: epidemiology and management of an alarming association. J Card Fail. 2008 Sep;14(7):615–25. - PubMed
-
- Boudina S, Abel ED. Diabetic cardiomyopathy revisited. Circulation. 2007 Jun 26;115(25):3213–23. - PubMed
-
- Zaid H, Antonescu CN, Randhawa VK, Klip A. Insulin action on glucose transporters through molecular switches, tracks and tethers. Biochem J. 2008 Jul 15;413(2):201–15. - PubMed
-
- Cantley LC. The phosphoinositide 3-kinase pathway. Science. 2002 May 31;296(5573):1655–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

