Smallpox vaccine with integrated IL-15 demonstrates enhanced in vivo viral clearance in immunodeficient mice and confers long term protection against a lethal monkeypox challenge in cynomolgus monkeys
- PMID: 20728526
- PMCID: PMC2952667
- DOI: 10.1016/j.vaccine.2010.08.008
Smallpox vaccine with integrated IL-15 demonstrates enhanced in vivo viral clearance in immunodeficient mice and confers long term protection against a lethal monkeypox challenge in cynomolgus monkeys
Abstract
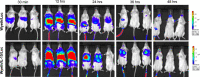
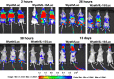
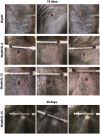
Despite the eradication of smallpox, there is heightened concern that it could be reintroduced as a result of intentional release of Variola major virus through an act of bioterrorism. The live vaccine that was pivotal in the eradication of smallpox though considered a gold standard for its efficacy still retains sufficient residual virulence that can cause life-threatening sequelae especially in immune deficient individuals. Therefore, a safer smallpox vaccine that can match the efficacy of first generation vaccines is urgently needed. We previously reported that the integration of human IL-15 cytokine into the genome of Wyeth strain of vaccinia (Wyeth/IL-15), the same strain as the licensed vaccine, generates a vaccine with superior immunogenicity and efficacy in a mouse model. We now demonstrate that Wyeth/IL-15 is non-lethal to athymic nude mice when administered intravenously at a dose of 10(7) plaque forming units and it undergoes enhanced in vivo clearance in these immune deficient mice. Furthermore, a majority of cynomolgus monkeys vaccinated with vaccinia viruses with integrated IL-15, when challenged 3 years later with a lethal dose of monkeypox virus displayed milder clinical manifestations with complete recovery supporting the utility of Wyeth/IL-15 for contemporary populations as a safer and efficacious smallpox vaccine.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Figures
References
-
- Fenner F., Henderson D.A., Arita I., Jecek Z., Ladnyi I.D. World Health Organization; Geneva, Switzerland: 1988. Smallpox and its eradication. History of International Public Health.
-
- Fenner F. Risks and benefits of vaccinia vaccine use in the worldwide smallpox eradication campaign. Res Virol. 1989;140:465–466. - PubMed
-
- Poland G.A., Grabenstein J.D., Neff J.M. The US smallpox vaccination program: a review of a large modern era smallpox vaccination implementation program. Vaccine. 2005;23:2078–2081. - PubMed
-
- Lane J.M., Ruben F.L., Neff J.M., Millar J.D. Complications of smallpox vaccination, 1968. N Engl J Med. 1969;281(22):1201–1208. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

